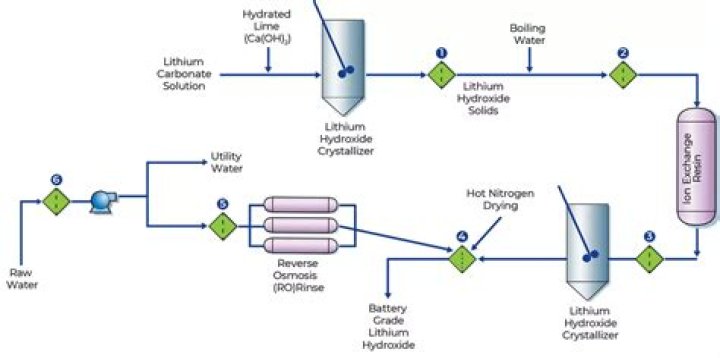
The name of the compound made from lithium and oxygen is d. lithium oxide.
Can a compound be made from lithium and oxygen?
The name of the compound made from lithium and oxygen is d. lithium oxide.
Is lithium oxide stable?
Lithium Oxide is a highly insoluble thermally stable Lithium source suitable for glass, optic and ceramic applications. Lithium oxide is a white solid also known as lithia, it is produced when lithium metal burns in the presence of oxygen. Oxide compounds are not conductive to electricity.
What compound does lithium and oxygen form?
The formula for lithium oxide is Li2 O. Lithium has one valence electron and loses it to form a lithium ion, Li+1. Oxygen gains two electrons to fill…How do you make a stable compound?
The octet rule states that an atom is most stable when there are eight electrons in its valence shell. Atoms with less than eight electrons tend to satisfy the duet rule, having two electrons in their valence shell. By satisfying the duet rule or the octet rule, ions are more stable.
What is the name of the compound made from lithium and oxygen quizlet?
Lithium and oxygen combine to form a compound called lithium oxide.
How does lithium become stable?
Lithium tends to lose one electron to take on the electron configuration of the nearest noble gas, helium, leaving it with two valence electrons. There are two ways in which atoms can satisfy the octet rule. One way is by sharing their valence electrons with other atoms.
Is lithium oxide a compound?
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). O) or lithia is an inorganic chemical compound. It is a white solid.What type of bond is formed between lithium and fluorine?
Ionic bonding: The formation of an ionic bond between lithium and fluorine to form LiF.
What is lithium oxygen?The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow. … In practice, Li–air batteries with a specific energy of ~6.12 MJ/kg at the cell level have been demonstrated.
Article first time published onIs Lithium oxide alkali?
Lithium oxide (Li2O) is the lightest alkali metal oxide and a white solid. It melts at 1570 °C.
Is Lithium oxide neutral?
a) Lithium oxide Li2O O = -2. The molecule is neutral, so the lithium atoms must have a total oxidation number of +2.
How is an ionic compound formed from lithium and oxygen?
Lithium oxide is an ionic compound formed between a metal (Li) and a non- metal (O) by the complete transfer of electrons from Li to O to give Li+ cations and O2– anions. These ions are held in place in the crystal lattice by strong electrostatic attractions between the positively and negatively charged ions.
How many electrons will lithium give up to be stable?
Recall that most atoms are stable when their outermost ring has eight electrons. (Some atoms, such as lithium and beryllium, are stable when their outermost ring has two electrons.)
What makes compounds more stable?
A molecule that has several resonance structures is more stable than one with fewer. Some resonance structures are more favorable than others.
What makes lithium unstable?
Lithium is a relatively unstable element because its stable isotopes have a very low binding energy level. This means they are not prone to fission, which is breaking down. This is why, lithium is less common in the galaxy than some other elements with a higher atomic weight.
How many oxygen atoms are stable?
Oxygen has six valence electrons in its outer shell. Eight electrons are needed to fill the second level to achieve stability. So oxygen needs two more electrons to be stable.
How can lithium achieve a noble gas configuration?
The noble gases are in Group 18 (VIIIA). Atoms of the representative elements achieve a noble gas configuration by gaining or losing electrons and becoming ions, or by sharing electrons. … After losing their valence electrons, lithium and beryllium ions will have the noble gas configuration of helium in Period 1.
Which of the following is an insoluble compound?
Explanation: “Insoluble” generally means that a substance does not dissolve in water. Some examples include: sand, fats, wood, metals, and plastic. When we put them in water and try to mix them, they will not dissolve.
What is the proper name for HBr AQ quizlet?
When dissolved in water, hydrogen bromide (HBr) forms hydrobromic acid.
Which compound is an example of a binary ionic compound?
A binary ionic compound is composed of ions of two different elements – one of which is a metal, and the other a nonmetal. For example, iron(III) iodide, FeI3, is composed of iron ions, Fe3+ (elemental iron is a metal), and iodide ions, I- (elemental iodine is a nonmetal).
Is lithium and fluorine stable after bonding?
The single electron in the outer shell of the Lithium atom jumps across and joins the seven electrons in the outer shell of the Fluorine atom. Lithium and Fluorine then have a complete outer shell of electrons. This is a more stable arrangement for each atom.
What happens when lithium and fluorine bond together?
Explanation: Lithium is an alkali metal and form an ionic bond by donating an electron. Fluorine is a halogen and forms ionic bonds by accepting an electron. Diagramming the formation of an ionic bond between lithium and fluorine looks exactly like the diagrammed bond between sodium and chlorine in the video below.
What happens when lithium and fluorine react to form an ionic bond?
Ionic bond: Electrons are transferred between atoms. … In the Figure below, an atom of lithium donates an electron to an atom of fluorine to form an ionic compound. The transfer of the electron gives the lithium ion a net charge of +1, and the fluorine ion a net charge of -1.
Is the oxygen molecule O2 a compound?
O2 is an compound . You might want to know why,Compound consists of elements that are bonded together to form a stable electron configuration. Now O2 consists of two atoms of oxygen which are covalently bonded so it is a compound.
Is oxygen a compound?
*Note: pure oxygen gas consists of molecules but it is still considered an element, rather than a compound, as the molecules are made up of a single type of element.
What type of oxide does lithium form?
Lithium (and to some extent sodium) form simple oxides, X2O, which contain the common O2− ion .
What happens when lithium and oxygen?
Lithium burns with a strongly red-tinged flame if heated in air. It reacts with oxygen in the air to give white lithium oxide. … For the record, it also reacts with the nitrogen in the air to give lithium nitride. Lithium is the only element in this Group to form a nitride in this way.
What is the balanced equation for lithium and oxygen?
How to Balance: Li + O2 = Li2O| Breslyn.org.
What is the name of the compound made from lithium and chlorine?
PubChem CID433294StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaClLi or LiClSynonymsLITHIUM CHLORIDE 7447-41-8 Lithium chloride (LiCl) Lithiumchloride LiCl More…
Does lithium form superoxide?
In a lithium-air battery, when there is a one electron reduction during discharge, lithium superoxide is formed as seen in the following reaction: Li+ + e− + O2 → LiO.