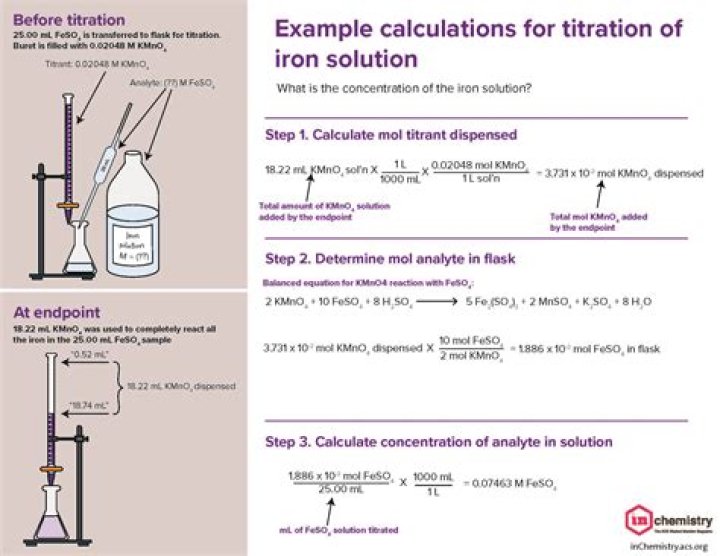
Typically, the titrant (the solution of known concentration) is added through a burette to a known volume of the analyte (the solution of unknown concentration) until the reaction is complete. Knowing the volume of titrant added allows us to determine the concentration of the unknown analyte.
How do you determine titrant and analyte?
Typically, the titrant (the solution of known concentration) is added through a burette to a known volume of the analyte (the solution of unknown concentration) until the reaction is complete. Knowing the volume of titrant added allows us to determine the concentration of the unknown analyte.
How do you find analyte concentration?
Use the titration formula. If the titrant and analyte have a 1:1 mole ratio, the formula is molarity (M) of the acid x volume (V) of the acid = molarity (M) of the base x volume (V) of the base. (Molarity is the concentration of a solution expressed as the number of moles of solute per litre of solution.)
What is analyte and titrant?
In analytical chemistry, the titrant is a solution of known concentration that is added (titrated) to another solution to determine the concentration of a second chemical species. … In contrast, the analyte, or titrand, is the species of interest during a titration.How do I calculate ka?
As noted above, [H3O+] = 10-pH. Since x = [H3O+] and you know the pH of the solution, you can write x = 10-2.4. It is now possible to find a numerical value for Ka. Ka = (10-2.4)2 /(0.9 – 10-2.4) = 1.8 x 10-5.
Is the titrant in the buret?
The titrant is added to the analyte using a precisely calibrated volumetric delivery tube called a burette (also spelled buret; see Figure 12.4. 1). The burette has markings to determine how much volume of solution has been added to the analyte. … This type of calculation is performed as part of a titration.
What is titrant chemistry?
Definition of titrant : a substance (such as a reagent solution of precisely known concentration) that is added in titration.
How do I calculate molar concentration?
To calculate the Molar Concentration, we will find the molar concentration by dividing the moles by liters of water used in the solution. For example, the acetic acid here is completely dissolved in 1.25 L of water. Then divide 0.1665 moles by 1.25 L to get the molar concentration, which will be 0.1332 M.How do you find the volume of a titrant?
Record the volume in the buret. This is your final volume (26.48 ml in this case). Subtract the initial volume (step 5) from the final volume to determine the volume of titrant added (26.48 – 14.62 = 11.76 ml).
What is an analyte test?An analyte is a substance or chemical constituent that is determined in an analytical procedure, such as a titration. … In medicine, analyte often refers to the type of test being run on a patient, as the test is usually determining a chemical substance in the human body.
Article first time published onWhat is meant by analyte in chemistry?
Definition of analyte : a chemical substance that is the subject of chemical analysis.
What is analyte solution?
The analyte (titrand) is the solution with an unknown molarity. The reagent (titrant) is the solution with a known molarity that will react with the analyte.
What is the difference between titrant and titrate?
is that titration is (analytical chemistry) the determination of the concentration of some substance in a solution by slowly adding measured amounts of some other substance (normally using a burette) until a reaction is shown to be complete, for instance by the colour change of an indicator while titrant is (analytical …
How do you calculate concentration from a titration curve?
Divide the number of moles of analyte present by the original volume of the analyte. For example, if the original volume of the analyte was 500 mL, divide by 1000 mL per L to obtain 0.5 L. Divide 0.01 moles of analyte by 0.5 L to obtain 0.02 moles per liter. This is the concentration or molarity.
What is the mole formula?
If you know the particles, moles, or grams of a substance, you can calculate the other two measurements by using the following equation: 1 mole = 6.022 × 1023 particles/mol = formula weight expressed in grams. From the Periodic Table, the mass of one atom of chromium is 51.9961 amu.
How do you calculate Ka from pKa?
To create a more manageable number, chemists define the pKa value as the negative logarithm of the Ka value: pKa = -log Ka. If you already know the pKa value for an acid and you need the Ka value, you find it by taking the antilog. In practice, this means raising both sides of the equality to exponents of 10.
What is titrant Mcq?
Titration or volumetric analysis is a method used in the laboratory to determine the concentration of an unknown solution. It is a quantitative method of chemical analysis. In this, a titrant of known concentration is prepared and used to react with the analyte to determine its concentration.
Is analyte in burette?
Typically, the titrant (the known solution) is added from a burette to the known quantity of the analyte (the unknown solution) until the reaction is complete.
Is the titrant always a base?
False; a base can be a titrant, or the reaction being performed may not even be an acid-base reaction.
What is meant by Standardisation in chemistry?
Standardization is the process of determining the exact concentration (molarity) of a solution. Titration is one type of analytical procedure often used in standardization. In a titration, an exact volume of one substance is reacted with a known amount of another substance.
Is molarity and concentration the same?
molarity: The concentration of a substance in solution, expressed as the number moles of solute per liter of solution. concentration: The relative amount of solute in a solution.
What is the initial Buret reading of titrant?
The initial reading on the buret was 2.74 mL.
How do you find concentration from molar mass and volume?
Concentration formula: To find the molar concentration of a solution, simply divide the total moles of solute by the total volume of the solution in liters.
What is analyte and sample?
sample: the object of the analytical procedure (for example: a blood sample); analyte: the substance that is of interest in the analysis (for example: amount of hemoglobin in blood); matrix: the constituents, apart from the analyte, of the given sample (for example: all the constituents of blood except hemoglobin);
What's analyte concentration?
(an’ă-līt) A material or substance the presence or concentration of which in a specimen is determined by analysis.
What are some examples of analytes?
- Alcohol.
- Ammonia.
- Ethanol.
- Glucose.
- Glutamine.
- Glycerol.
- Lactate.
- Lactose.
What is analyte and matrix?
An analysis provides chemical or physical information about a sample. The component in the sample of interest to us is called the analyte, and the remainder of the sample is the matrix. In an analysis we determine the identity, the concentration, or the properties of an analyte.
What is analyte in biology?
Analyte: A substance of interest that needs detection. For instance, glucose is an ‘analyte’ in a biosensor designed to detect glucose. Bioreceptor: A molecule that specifically recognises the analyte is known as a bioreceptor.
What is the analyte in chromatography?
Chromatography terms. Analyte – the substance to be separated during chromatography. It is also normally what is needed from the mixture. Analytical chromatography – the use of chromatography to determine the existence and possibly also the concentration of analyte(s) in a sample.
Is NaOH an analyte?
The most common use of titrations is for determining the unknown concentration of a component (the analyte) in a solution by reacting it with a solution of another compound (the titrant). … During the course of the titration, the titrant (NaOH) is added slowly to the unknown solution.
Is the analyte titrated?
The titrant reacts with a solution of analyte (which may also be termed the titrand) to determine the analyte’s concentration. The volume of titrant that reacted with the analyte is termed the titration volume.