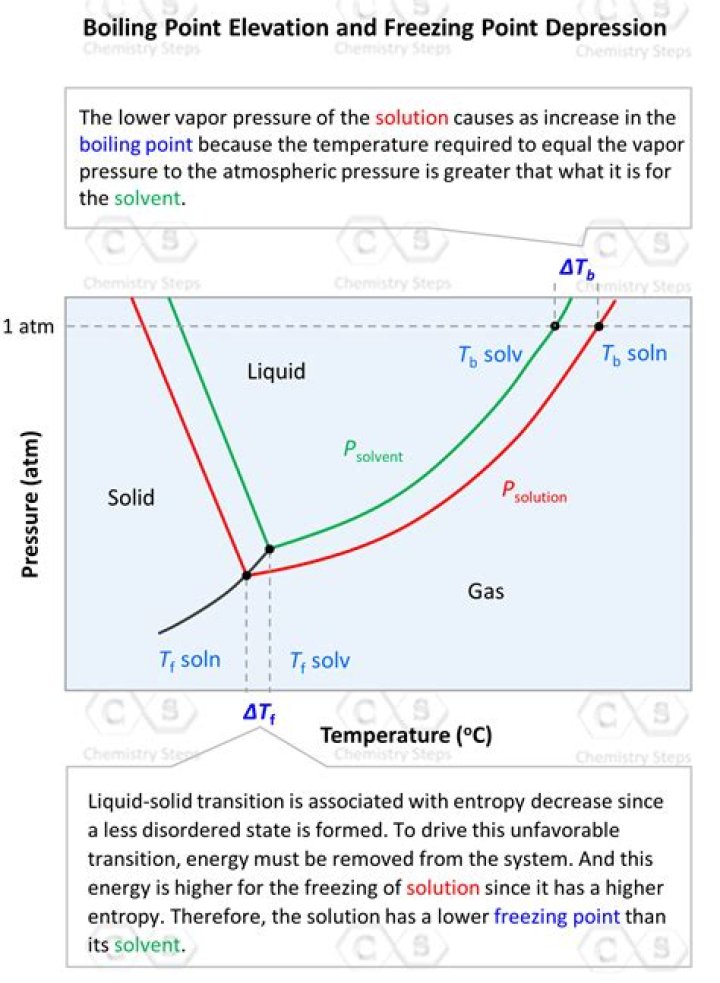
Freezing and Boiling. Lowering the vapor pressure of a substance has an obvious effect on boiling point; the boiling point goes up. … Thus, the temperature of the boiling point must be increased. Another consequence of lowered vapor pressure is a decrease in freezing point.
Why does freezing point decrease when pressure increases?
We know that with increasing atmospheric pressure, the freezing point of water slightly decreases. Because of increasing the pressure, which prevents water molecules properly arranging them. If water freezes, the mean forms a crystalline solid phase with a proper arrangement. … That means water freezes at below 273K.
Is freezing affected by pressure?
Most importantly, changes in the pressure can increase or decrease the freezing point of a substance. Typically, pressure lower than 1atm reduces the temperature at which the substance freezes. But in the case of water, the higher the pressure, lower is the freezing point.
What happens to the vapor pressure boiling point and freezing point?
Freezing point decreases, boiling point increases, and vapor pressure decreases. Whenever ANYTHING at all is added to a solvent, it blocks some solvent particles at the surface of the solution, or attracts solvent particles towards it.What affects the freezing point of a solution?
The freezing point of the solvent in a solution changes as the concentration of the solute in the solution changes (but it does not depend on the identity of either the solvent or the solute(s) particles (kind, size or charge) in the solution).
How does vapor pressure affect melting point?
Increasing the atmospheric pressure will generally increase the melting point of most substances, therefore increasing the energy required to facilitate fusion. This is because of volume: Most solids have less volume than liquids, so melting would cause the solid to expand.
How does lower pressure affect melting point?
if we will increase the pressure they will be needed more energy to melt and if we decrease the pressure the amount of energy required to get the atoms of that solid excited will be less. Assuming that we are talking about water- with an increase in pressure, the melting point of water reduces.
Why is vapour pressure inversely proportional to boiling point?
– Whereas, if the intermolecular forces are weak, then liquid will have high vapour pressure, little amount of energy will be added to separate the molecules. And hence the boiling point will be low. – Hence, we can say that vapour pressure and boiling point are inversely proportional to each other.What is the effect of vapour pressure on boiling point?
The higher the vapor pressure of a liquid at a given temperature, the lower the normal boiling point (i.e., the boiling point at atmospheric pressure) of the liquid. The vapor pressure chart to the right has graphs of the vapor pressures versus temperatures for a variety of liquids.
What increases freezing point?freezing point, temperature at which a liquid becomes a solid. As with the melting point, increased pressure usually raises the freezing point. The freezing point is lower than the melting point in the case of mixtures and for certain organic compounds such as fats.
Article first time published onHow do intermolecular forces affect freezing point?
Molecules with stronger intermolecular forces are pulled together tightly to form a solid at higher temperatures, so their freezing point is higher. Molecules with lower intermolecular forces will not solidify until the temperature is lowered further.
What does vapor pressure lowering mean?
Vapor pressure lowering is a colligative property of solutions. The vapor pressure of a pure solvent is greater than the vapor pressure of a solution containing a non volatile liquid. This lowered vapor pressure leads to boiling point elevation. vapor pressure lowering.
Does vapor pressure increase with solute?
As more solute is dissolved in the solvent, the vapor pressure of the solvent decreases, and the change in the vapor pressure of the solvent increases.
Does pressure increase melting point of ice?
The correct answer is Decreases. With the increase in pressure, the melting point of ice decreases because with increased pressure the volume decreases.
How does pressure affect boiling and melting point?
Atmospheric pressure influences the boiling point of water. When atmospheric pressure increases, the boiling point becomes higher, and when atmospheric pressure decreases (as it does when elevation increases), the boiling point becomes lower.
How does pressure affect ice?
In this case, if you increase the pressure on the ice the ice-water system wants to try to lower it again. It can do that by making itself fit into a smaller volume. But since water fills a smaller volume when it’s liquid, rather than solid, it will go to a lower melting point — allowing more solid to become liquid.
Does vapor pressure increase with temperature?
As the temperature of a liquid increases, the kinetic energy of its molecules also increases and as the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure.
Does pressure increase melting point?
Most liquids are less dense than the solid phase, so higher pressure increase the melting point. The dotted green line shows the melting point for water. Water is denser as a liquid, so higher pressures decrease the melting temperature.
Is vapor pressure proportional to temperature?
The vapor pressure of a liquid varies with its temperature, as the following graph shows for water. … As the temperature of a liquid or solid increases its vapor pressure also increases. Conversely, vapor pressure decreases as the temperature decreases.
What are the factors affecting vapour pressure?
Three common factors that influence vapor press are surface area, intermolecular forces and temperature. The vapor pressure of a molecule differs at different temperatures.
What is the difference between vapour pressure and boiling point?
Boiling causes the phase change of a liquid into its vapor. Vaporization gives rise to the vapor pressure of a closed system. … The main difference between vapor pressure and boiling point is that vapor pressure is a measurement of pressure whereas boiling point is a measurement of temperature.
Does high boiling point mean low vapor pressure?
Vapor pressure and boiling point are both dependent on intermolecular forces and temperature. The greater the intermolecular forces, the higher the boiling point and lower the vapor pressure.
What is the relation between boiling point and vapour pressure and evaporation?
The vapour pressure of the liquid will be high if the intermolecular forces are weak. In this case, very less heat energy is required to separate the molecules. Therefore, the boiling point will be low. When the intermolecular force is strong, then the vapour pressure will be low and the boiling point will be high.
What causes a lower freezing point?
Freezing point depression is the phenomena that describes why adding a solute to a solvent results in the lowering of the freezing point of the solvent. When a substance starts to freeze, the molecules slow down due to the decreases in temperature, and the intermolecular forces start to take over.
How does Molality affect freezing point?
The depression in freezing point is directly proportional to the molality of the solution where the molality is the number of solute dissolved in one kilogram of the solvent. … So, when more concentrated will be the solution in other words more number of solute particles, the freezing point will be depressed more.
What lowers the freezing point of water?
The two most common ingredients in cooking that affect freezing point are salt and sugar. Sugar lowers the freezing point of water, which makes frozen desserts fair game for changes in freezing point. Most desserts freeze between 29.5 to 26.6 degrees F (-1.4 to -3.0 C) depending on the concentration of sugar.
How is vapor pressure affected by intermolecular forces?
A liquid’s vapor pressure is directly related to the intermolecular forces present between its molecules. The stronger these forces, the lower the rate of evaporation and the lower the vapor pressure.
Does volume affect freezing point?
The freezing point depends on hydrostatic pressure depending on the change in volume between solid and liquid. The more dense phase is favored by higher pressure. If the hydrostatic pressure is an inert gas the vapor pressures of the solid and liquid are equal at the melting-freezing temperature.
What happens to the particles during freezing?
Freezing occurs when a liquid is cooled and turns to a solid. Eventually the particles in a liquid stop moving about and settle into a stable arrangement, forming a solid. This is called freezing and occurs at the same temperature as melting.
What is the importance of vapor pressure?
The vapour is said to be “pushing” against the atmosphere. In other words, the higher the vapour pressure the faster a liquid evaporates. When the vapour pressure reaches the atmospheric pressure, the liquid is at its boiling point.
What does vapor pressure lowering depend on?
The lowering of the vapor pressure depends on the number of solute particles that have been dissolved. The chemical nature of the solute is not important because the vapor pressure is merely a physical property of the solvent.