List the member elements by their symbol. There are 5 elements belong to the Halogen family. They are F, Cl, Br, I, and At. What type of molecule is formed when a halogen combines with hydrogen?
Which are the elements that belong to halogen family?
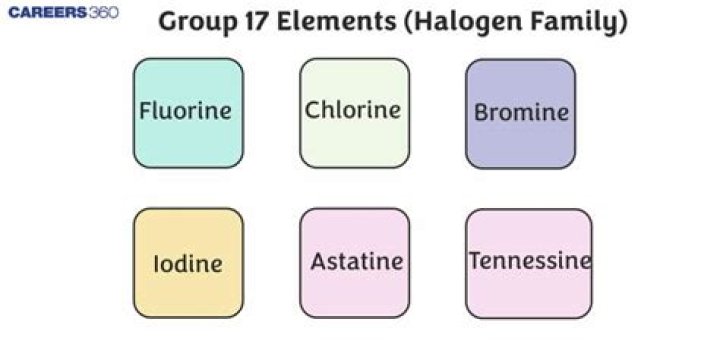
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
What do the elements in the halogen family have in common?
Summary of Common Properties They have seven valence electrons (one short of a stable octet). They are highly reactive, especially with alkali metals and alkaline earths. Halogens are the most reactive nonmetals. Because they are so reactive, elemental halogens are toxic and potentially lethal.
What are Group 7 halogens?
The Group 7 elements are called the halogens. They are placed in the vertical column, second from the right, in the periodic table . Chlorine, bromine and iodine are the three common Group 7 elements. Group 7 elements form salts when they react with metals.How many elements belong to the alkali metal family list the member elements by their symbol?
The alkali metals are six chemical elements in Group 1, the leftmost column in the periodic table. They are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
Which of the following elements belong to halogen family Brainly?
The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
Why are 17 elements called halogens?
The group 17 elements include fluorine(F), chlorine(Cl), bromine(Br), iodine(I) and astatine(At) from the top to the bottom. They are called “halogens” because they give salts when they react with metals.
What are the Group 6 elements called?
Group 6A (or VIA) of the periodic table are the chalcogens: the nonmetals oxygen (O), sulfur (S), and selenium (Se), the metalloid tellurium (Te), and the metal polonium (Po).What are the Group 8 elements called?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn).
What are the Group 0 elements?The Group 0 elements, the NOBLE GASES consist of helium, neon, argon, krypton, xenon and radioactive radon.
Article first time published onWhat does Group 17 have in common?
Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They have seven valence electrons, so they are very “eager” to gain one electron to have a full outer energy level. Most halogens have a variety of important uses, such as preventing tooth decay and killing germs.
What do group 17 elements become when they react with group 1 elements?
The elements of Group VIIA (new Group 17 – fluorine, chlorine, bromine, iodine, and astatine) are called the halogens (tan column). The term “halogen” means “salt-former” because these elements will readily react with alkali metal and alkaline earth metals to form halide salts.
What are the Group 18 elements called?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
When an alkali metal reacts with a halogen?
Exposing an alkali metal to a halogen will cause an extremely exothermic reaction that results in an ionic salt. Almost every salt of an alkali metal is highly soluble in water. They form conducting solutions, proving their ionic nature.
What are the elements belongs to alkali metals?
Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). These are (except for hydrogen) soft, shiny, low-melting, highly reactive metals, which tarnish when exposed to air.
What are alkali metals Class 10?
In the periodic table, the alkali metals are a group or column containing the chemical elements such as lithium (Li), sodium (Na), rubidium (Rb), potassium (K), francium (Fr) and Caesium (Cs). … All the alkali metals react with water, with the heavier alkali metals reacting more overwhelmingly than the lighter ones.
What are halogens Class 9?
Halogens are nonmetals. At room temperature, fluorine and chlorine are gases and bromine is a liquid. Halogens are very reactive, the reactivity decreases from fluorine to astatine. …
Why are group 16 called halogens?
They are called “halogens” because they give salts when they react with metals. The valence shell electronic configuration of these electrons is ns2np5. … The electron gain enthalpy of these elements becomes less negative upon moving down the group. Fluorine has lesser enthalpy than chlorine.
Why are halogens known as Group 7 elements?
Group 7 elements form salts when they react with metals. The term ‘halogen’ means ‘salt former’, which is why Group 7 elements are called halogens. In general the halogens comprise the most reactive group of non-metals. The halogens are so reactive that they cannot exist free in nature.
What is the 32nd element?
Germanium – Element information, properties and uses | Periodic Table.
Which of the following is NOT belong to halogen family?
Detailed Solution. The correct answer is option 4, i.e. Zirconium. Halogen, any of the six nonmetallic elements that constitute Group 17 of the periodic table. It is not a halogen element.
Which one of the following elements show the highest electronegativity?
Thus, fluorine is the most electronegative element, while francium is one of the least electronegative. (Helium, neon, and argon are not listed in the Pauling electronegativity scale, although in the Allred-Rochow scale, helium has the highest electronegativity.)
What are group 9 elements called?
IUPAC group number9Name by elementcobalt groupCAS group number (US, pattern A-B-A)part of VIIIBold IUPAC number (Europe, pattern A-B)part of VIII
What family is Group 8?
New IUPAC nameOld IUPAC (Europe)Name by elementGroup 8VIIIiron familyGroup 9VIIIcobalt familyGroup 10VIIInickel familyGroup 11IBcopper family
What element is in group 1 Period 8?
Group 8 is a group (column) of chemical elements in the periodic table. It consists of iron (Fe), ruthenium (Ru), osmium (Os) and hassium (Hs). They are all transition metals.
What is the name of group 16 elements?
oxygen group element, also called chalcogen, any of the six chemical elements making up Group 16 (VIa) of the periodic classification—namely, oxygen (O), sulfur (S), selenium (Se), tellurium (Te), polonium (Po), and livermorium (Lv).
What does the elemental symbol V mean?
Vanadium is a chemical element with symbol V and atomic number 23.
What are the group 5 elements called?
IUPAC group number5Name by elementvanadium groupCAS group number (US, pattern A-B-A)VBold IUPAC number (Europe, pattern A-B)VA
What are Group 7 elements called?
Group 7A (or VIIA) of the periodic table are the halogens: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The name “halogen” means “salt former”, derived from the Greek words halo- (“salt”) and -gen (“formation”).
What is Group 7 on the periodic table called?
Group 7 Elements Also called the halogens. molecules that contain two atoms.
What is Group 1 on the periodic table called?
The Group 1 elements are called the alkali metals. They are placed in the vertical column on the left-hand side of the periodic table . All the Group 1 elements are very reactive .