The density of the dichloromethane is greater than that of the water, therefore it sinks to the bottom.
Why does water and dichloromethane separate?
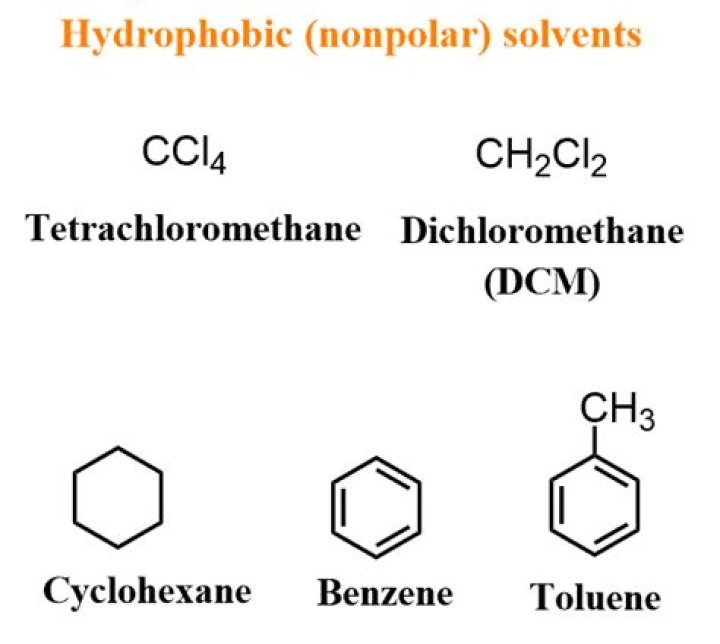
Immiscibility. Organic solvents like dichloromethane are immiscible in water because water is, by contrast, a very polar solvent. … However, DCM is actually denser than water, and leaves an organic layer below the aqueous layer rather than above like other solvents.
Which has a higher density water or methylene chloride?
Whether the water layer is on the top or bottom depends on the density of the other solvent (methylene chloride is heavier than water and goes to the bottom of the separtory funnel and diethyl ether and ethyl acetate are lighter than water and stay on the top).
What fluids are more dense than water?
Glycerol (or Glycerin) is more dense than water (1.26 g/cc). One could argue that glass is a very slow-moving, viscous liquid (although it has lots of properties of a solid, like rigidity). It’s more dense than water. Even saltwater is more dense than water.Does dichloromethane float in water?
The density of the dichloromethane is greater than that of the water, therefore it sinks to the bottom.
Is dichloromethane denser or less dense than water explain your reasoning?
Chlorinated solvents (i.e., dichloromethane, chloroform) exhibit a higher density than water, while ethers, hydrocarbons and many esters possess a lower density than water (see solvent table), thus form the top layer (see solvent table)..
Does dichloromethane dissolve in water?
It is moderately soluble in water (2 g/100 ml at 20 °C) and soluble in most organic solvents such as ethanol, ether, phenols, aldehydes and ketones. Its evaporation rate is 27.5 (reference liquid is butyl acetate = 1). DCM vapours are heavier than air.
Which liquid is the least dense?
What is the least dense liquid in the world? – Quora. For liquids which are commonly found in everyday life, gasoline has a pretty low density: 0.7 g/ml. It is about 30% lighter than water which has a density of 1.0 g/ml. Compared to liquid hydrogen, however, gasoline is about 10x heavier.What are the densities of water diethyl ether and dichloromethane?
The density of dichloromethane, an organic solvent, is 1.325 g/cm3 . If you mix 50 mL water and 50 mL dichloromethane together in a separatory funnel, which layer would be on top? The density of diethyl ether, an organic solvent, is 0.706 g/cm3 .
What's the most dense liquid?Mercury is the densest liquid at standard conditions for temperature and pressure (STP). Also called quicksilver, mercury has been known for more than 3,500 years. It is an important metal in industry, but it is also toxic.
Article first time published onWhich gas is more dense?
Densities of Common Elements and CompoundsSubstanceDensity grams per mLHydrogen gas0.000089Helium gas0.00018Air0.00128
Which solvent is denser than water?
The only common solvent that is denser than water is Dichloromethane (DCM). Because density partially depends on molecular weight, the two chlorine atoms, which have a high molecular weight, make it denser than water.
Which substance has less density than water?
Even though it’s heavier, wax has a lower density than water, so the big candle floats. Sinking and floating applies to liquids too. For example, if you add vegetable oil to water, the oil floats on top of the water because the oil has a lower density than the water.
Is hexane more dense than water?
b; hexane is insoluble in water and less dense than water.
Is cyclohexane more dense than water?
cyclohexane is less dense than water.
Why is water more dense?
At 39°F (or 3.98°C to be exact) water is the most dense. … This is because the molecular structure and hydrogen bonding of the water takes up more space when it is a frozen solid rather than when it is a warmer liquid. In other words, the molecules are farther part even though the water is solidifying into ice.
How is dichloromethane removed from water?
Air stripping, adsorption and pervaporation were found to be effective in separating DCM from water with a process efficiency of about 99%, 90% and 80% respectively.
Is dichloromethane polar or non polar?
Still, Dichloromethane, also known as Methyl Chloride, develops a net dipole moment across C-Cl and C-H bonds. The chemical bond results in a net 1.67 D dipole moment, thus making it a polar compound.
Can dichloromethane hydrogen bond with water?
Dichloromethane and propane contain hydrogen, but they don’t contain nitrogen, oxygen, or fluorine; therefore, they can’t form hydrogen bonds.
Is diethyl ether more dense than water?
Diethyl ether appears as a clear colorless liquid with an anesthetic odor. Flash point -49°F. Less dense than water and slightly soluble in water.
Is benzoic acid soluble in dichloromethane?
Caffeine and benzoic acid are both soluble in DCM, but cellulose is not.
Why does benzoic acid dissolve in dichloromethane?
Benzoic acid is a polar protic compound due to the presence of the polar protic -COOH group. … Hence, it is expected that benzoic acid to be soluble in both water dichloromethane by means of polar intermolecular forces, e.g., dipole-dipole interaction and hydrogen bonding.
What are some dense liquids?
LiquidTemperature – t – (oC)Density – ρ – (kg/m3)Alcohol, ethyl (ethanol)25785.1Alcohol, methyl (methanol)25786.5Alcohol, propyl25800.0Almond kernel oil25910
What is the density of water in cubic centimeters?
A common unit of measurement for water’s density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm3). Actually, the exact density of water is not really 1 g/ml, but rather a bit less (very, very little less), at 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit).
Is dichloromethane organic or aqueous?
Typical organic solvents include ethyl acetate, hexane, chloroform, dichloromethane, and diethyl ether. All of these form a crisp delineation between the two liquids. The two layers are commonly referred to as the aqueous phase and the organic phase.
Which solvent has the highest density?
SolventFormuladensity (g/mL)acetic acidC2H4O21.0446acetoneC3H6O0.7845acetonitrileC2H3N0.7857benzeneC6H60.8765
Which is more dense methanol or hexane?
Hexane is less dense than either methanol or water, so floats on top.
Are gasses dense?
And the answer is yes. A gas can be more dense than a liquid. … The density of a gas is dependent upon the pressure and the temperature that it is under. The density of a gas is directly proportional to the pressure and indirectly proportional to the temperature.
Are there any liquids less dense than water?
MaterialDensity (g/cm3)Water1.00Ice Cube0.92Vegetable Oil0.92Baby Oil0.83
Why is glycerol more dense than water?
Well, glycerine, 1,2,3-propane-triol, has more extensive opportunity for intermolecular hydrogen-bonding, and this elevates the density, as well as the boiling point. Glycerine has higher melting point, and (much) higher boiling point than water, consistent with a grea…
Which solid has highest density?
In solids, Os has the highest density.