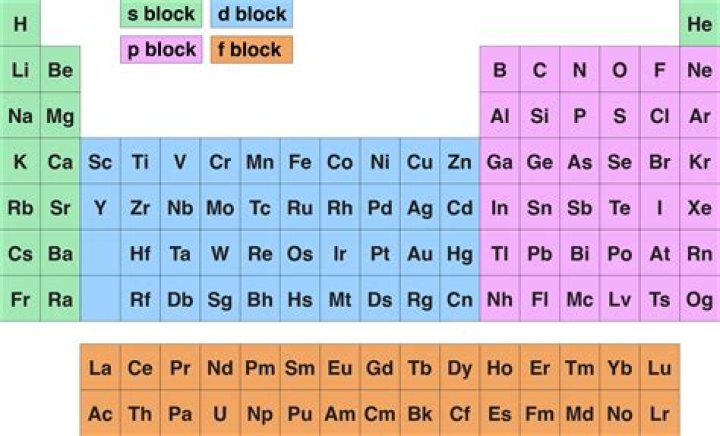
Periodic table blocks are sets of elements grouped by their valence electron orbitals. The four block names are s-block, p-block, d-block, and f-block
What do the blocks represent on the periodic table?
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-block, p-block, d-block, and f-block.
What do the 7 rows on the periodic table represent?
The table starts with the simplest atom, hydrogen, and then organizes the rest of the elements by atomic number, which is the number of protons each contains. … The table has seven rows and 18 columns. Each row represents one period; the period number of an element indicates how many of its energy levels house electrons.
What are the 4 main blocks of the periodic table?
This gives the periodic table block in the standard periodic table in which the element is to be found. The four blocks are s, p, d, and f. Elements in each block are colour coded in the background graphics, the periodic tables, and the element pages themselves.How many blocks are in the periodic table?
On the basis of electronic configuration, the elements have been categorized into four blocks in the periodic table. Complete step by step answer: On the basis of electronic configurations, the elements in the long form periodic table have been divided into four blocks. These are s, p, d, f-blocks.
What is each block in a table called?
Answer: The element blocks are s, p, d, and f. … The four block names are s-block, p-block, d-block, and f-block.
How are blocks indicated?
Summary. The horizontal rows of the periodic table are called periods. The length of a period depends on how many electrons are needed to occupy the sublevels that fill the period. Blocks indicate which sublevel is being filled.
Why does d-block Start 3?
If you do the math you’ll see that in the 3rd period there’s the main quantum number n=3 which allows the secondary quantum number l to take the values 0, 1 or 2. As l determines the kind of the orbital you get a s, p or d orbital.Where is the f-block on the periodic table?
The f-block elements,found in the two rows at the bottom of the periodic table, are called inner transition metals and have valence electrons in the f-orbital’s.
How do you read block elements?Element Boxes The atomic number, the number at the top of each element box, is the number of protons in the nucleus and the number of electrons in the atom. The elemental symbol is an abbreviation of the element name. Some periodic tables do not include the element names, only the symbols.
Article first time published onWhat blocks of the periodic table make up representative elements?
The elements in the periodic table are arranged into four blocks: s, p, d, and f. The elements present in s- and p-blocks are collectively known as representative elements or main group elements.
Which element is in group 14 Period 3?
Silicon (symbol Si) is a group 14 metalloid.
How many blocks are there in periodic table write main features of each block?
The modern periodic table is divided into 4 blocks i.e s, p, d and f block.
Why are there 15 elements in the f-block?
If your periodic table shows 15, then one of the elements in that block represents the one slot of the d-block that is left vacant when indicating the place the f-block “inserts.” Note that in the rows above those, there is one element in that slot. So it’s 14 f-block slots plus one d-block slot.
What is the basis of dividing periodic table in blocks?
Elements in the long form of periodic table have been divided into four blocks i.e. s ,p ,d and f. This division is based upon the name of the orbitals which receives the last electron. 1)Elements in which the last electron enters the s orbital of their respective outermost shells are called s block elements.
In which block of the modern periodic table are the nonmetals found answer?
Non-metals are found in the p−block of the modern periodic table.
How many columns are in the S block?
How many columns are in the s block? There are two columns in the s block. Each s sublevel can have a total of 2 electrons.
What block is iron Fe?
Group81538°C, 2800°F, 1811 KBlockd7.87Atomic number2655.845State at 20°CSolid56FeElectron configuration[Ar] 3d64s27439-89-6
Why is it called the f-block?
(Public Domain). The f block elements are the lanthanides and actinides and are called the inner transition elements because of their placement in the periodic table due to their electron configurations.
Why is it called 3d and not 4d?
As you can see, the 4S orbital is filled BEFORE the 3D orbital as it has a lower energy, and therefore 3D has to be placed in the 4th row in the periodic table, after 4S. This trend continues as you go down the rows. For example, the 5th row starts with 5S and is then followed by 4D, and so on. Hope this helps!
Why is the D block N 1?
The (n–1) remains for the inward d orbitals which may have one to ten electrons and the peripheral ns orbital may have one or two electrons. … The d–orbitals of the penultimate energy level in their atoms get electrons leading to the three columns of the transition metals, i.e., 3d, 4d and 5d.
Does Period 3 have d orbital?
As mentioned above, all elements in period 3 have access to their d-orbitals because their principal quantum number n=3. With n=3, the angular quantum number l can be 0, 1, and 2, which corresponds to s, p, and d-orbitals respectively.
What does 3B mean on the periodic table?
Group 3B (3) The Group 3B elements (Group 3 in the IUPAC designation) usually have electron configuration (n-1)d1 ns2.
What does group A and B mean on the periodic table?
Elements are arranged by reactivity in the periodic table. Elements with similar reactivity are put into the same column or group. The columns with B (IB through VIIIB) are called the transition elements. … The columns with A (IA through VIIIA) are called the main group elements.
Which blocks represent the transition elements?
Many scientists describe a “transition metal” as any element in the d-block of the periodic table, which includes groups 3 to 12 on the periodic table. In actual practice, the f-block lanthanide and actinide series are also considered transition metals and are called “inner transition metals”.
Which block is inner transition elements?
Inner transition metals are in the f-block and have valence electrons in the f-orbital’s. They consist of the Lanthanides and the Actinides.
What is representative element?
The representative elements are elements where the s and p orbitals are filling. The transition elements are elements where the d orbitals (groups 3–11 on the periodic table) are filling, and the inner transition metals are the elements where the f orbitals are filling.
What element is in period 4 Group 18?
PeriodGroup3Na 11Ar 184K 19Kr 365Rb 37Xe 546Cs 55Rn 86
What element is in period 4 Group 11?
Copper. Copper (Cu) is an element in group 11.
Which element is in group 12 Period 4?
zinc group element, any of the four chemical elements that constitute Group 12 (IIb) of the periodic table—namely, zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn).
What is the block in the middle of the periodic table called?
d-block. The d-block is in the middle of the periodic table. Elements from groups 3 to 12 are in the d-block.