1 : characterized by or formed with absorption of heat. 2 : warm-blooded.
What does exothermic mean in science?
Definition of exothermic : characterized by or formed with evolution of heat.
What does endothermic mean example?
The definition of endothermic is a chemical reaction that is accompanied by the absorption of heat, or an organism that generates heat to maintain its temperature. … A warm-blodded animal would be an example of an endothermic animal.
What is the meaning of endothermic reaction in science?
Definitions of endothermic reaction. a chemical reaction accompanied by the absorption of heat. type of: chemical reaction, reaction. (chemistry) a process in which one or more substances are changed into others.What is exothermic and endothermic in science?
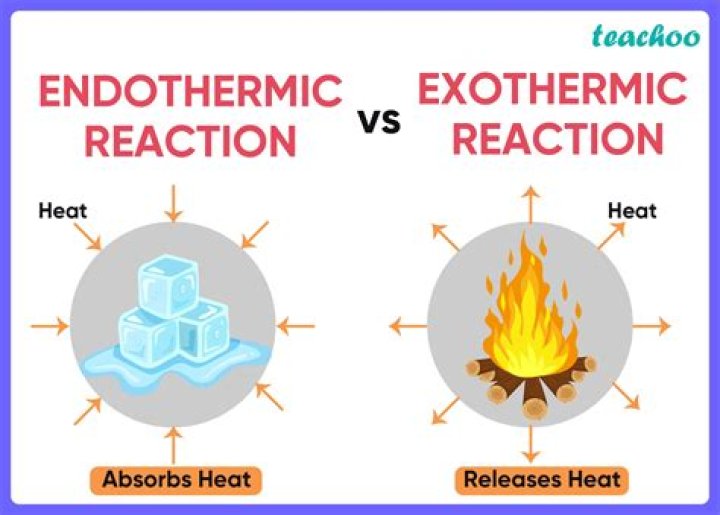
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.”
Is endothermic positive or negative?
So, if a reaction releases more energy than it absorbs, the reaction is exothermic and enthalpy will be negative. Think of this as an amount of heat leaving (or being subtracted from) the reaction. If a reaction absorbs or uses more energy than it releases, the reaction is endothermic, and enthalpy will be positive.
Is photosynthesis exothermic or endothermic?
Photosynthesis is an endothermic reaction. This means it cannot occur without energy (from the Sun). The light required is absorbed by a green pigment called chlorophyll in the leaves.
What are 5 examples of endothermic reactions?
- Melting ice cubes.
- Melting solid salts.
- Sublimation of dry ice into carbon dioxide gas.
- Evaporating liquid water.
- Converting frost to water vapor (melting, boiling, evaporation, and sublimation are endothermic processes)
- Making an anhydrous salt from a hydrate.
What are 2 examples of endothermic reactions?
- Melting ice cubes.
- Melting solid salts.
- Evaporating liquid water.
- Converting frost to water vapor (melting, boiling, and evaporation, in general, are endothermic processes.
- Making of an ice cube. Making an ice cube is a process of liquid changing its state to solid. …
- Snow formation in clouds. …
- Burning of a candle. …
- Rusting of iron. …
- Burning of sugar. …
- Formation of ion pairs. …
- Reaction of Strong acid and Water. …
- Water and calcium chloride.
How are reactions endothermic?
Chemical reactions that absorb (or use) energy overall are called endothermic. In endothermic reactions, more energy is absorbed when the bonds in the reactants are broken than is released when new bonds are formed in the products.
What do you mean by exothermic and endothermic reaction give Example Class 10?
Give examples. Answer: The chemical reactions which occur with the evolution of heat are called exothermic reactions. For example, Heat Heat The chemical reactions which occur with the absorption of heat are called endothermic reactions.
What event is endothermic?
In thermochemistry, an endothermic process is any process with an increase in the enthalpy H (or internal energy U) of the system. In such a process, a closed system usually absorbs thermal energy from its surroundings, which is heat transfer into the system.
Why photosynthesis is called endothermic?
Photosynthesis is an endothermic reaction because sunlight energy is absorbed by green plants during this process.
Why is photosynthesis endothermic?
Plants make their own food using photosynthesis . … Photosynthesis is an endothermic reaction as it requires light energy to react carbon dioxide and water to produce glucose and oxygen. The light energy required is absorbed by a green pigment called chlorophyll in the leaves.
Why photosynthesis is called as an endothermic reaction?
Photosynthesis is considered an endothermic reaction, because during the process of photosynthesis, energy from the sun or sunlight is being absorbed. Any chemical reactions that absorb heat energy from the surroundings to form products is termed the endothermic reaction.
What does Delta's mean?
Delta S is entropy. It’s a measurement of randomness or disorder. Notice I have deltas in front of these. That’s because we typically talk about changes, reactions or processes that actually happen in Chemistry. … We can only measure the change it undergoes through a chemical process.
Is endothermic hot or cold?
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.”
What does Delta u mean in chemistry?
Here Δ U \Delta U ΔU is the change in internal energy U of the system. Q Q. Q is the net heat transferred into the system—that is, Q is the sum of all heat transfer into and out of the system. W W.
Which reaction is the most endothermic?
The most endothermic reactions are entropy driven. In order to absorb heat from the environment, energy must be redistributed in an even less available form than random energy at that temperature. This means it must be given more degrees of freedom, the freedom of more molecular states than before.
Is a reaction endothermic or exothermic?
Endothermic ReactionsExothermic ReactionsHeat is absorbed by reactants to form products.Heat is released.Heat is absorbed from the surroundings; as a result, the surroundings get cold.Heat is released by the reaction to surroundings; surroundings feel hot.ΔHrxn is positiveΔHrxn is negative
Which reactions are exothermic?
- The thermite reaction.
- A neutralization reaction (e.g., mixing an acid and a base to form a salt and water)
- Most polymerization reactions.
- Combustion of a fuel.
- Respiration.
- Nuclear fission.
- Corrosion of metal (an oxidation reaction)
- Dissolving an acid in water.
What is an example of a biochemical reaction?
Types of Biochemical Reactions These reactions break down molecules into smaller units and release energy. An example of a catabolic reaction is the breakdown of glucose, which releases energy that cells need to carry out life processes. Endothermic reactions in organisms are called anabolic reactions.
What are 3 exothermic reactions?
- any combustion reaction.
- a neutralization reaction.
- rusting of iron (rust steel wool with vinegar)
- the thermite reaction.
- reaction between water and calcium chloride.
- reaction between sodium sulfite and bleach (dilute sodium hypochlorite)
- reaction between potassium permanganate and glycerol.
Which of the following is an endothermic process?
Fusion, vaporization, and sublimation are endothermic processes.
What are the examples of exothermic and endothermic reaction?
Endothermic ReactionExothermic ReactionEnergy in the form of heatEnergy is released as heat, electricity, light or sound.Melting ice, evaporation, cooking, gas molecules, photosynthesis are a few examplesRusting iron, settling, chemical bonds, explosions, nuclear fission are a few examples.
What are some examples of endothermic and exothermic reactions that you see in everyday life?
Exothermic processesEndothermic processescondensation of rain from water vaporevaporation of watera candle flameforming a cation from an atom in the gas phasemixing sodium sulfite and bleachbaking breadrusting ironcooking an egg
Is a hot pack endothermic?
A hot pack is produced if an exothermic reaction occurs as the salt and water mix and heat energy is released because this process will raise the temperature of the contents in the pack.
What does one mean by exothermic and endothermic reactions give examples 3?
The chemical reactions that release heat energy are called exothermic reactions. Example: C(g)+O2(g)→CO2(g)+Heat Energy. The chemical reactions in which heat energy is absorbed are called endothermic reactions. Example: CaCO3Heat→CaO+CO2.
What is endothermic reaction Class 10th?
Endothermic reactions are chemical reactions in which the reactants absorb heat energy from the surroundings to form products.
What does one mean by exothermic and endothermic reactions give one example each?
Exothermic reactions : Reactions in which heat is given out along with the products are called exothermic reactions. Example : Endothermic reactions:Reactions in which energy is absorbed are known as endothermic reactions. Example : The process of photosynthesis.