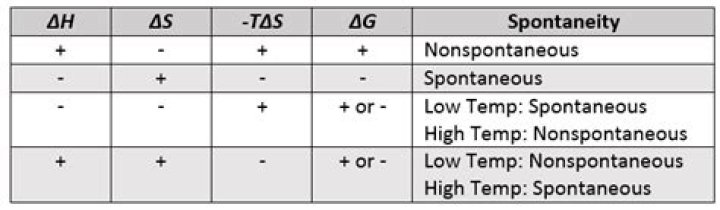
Entropy ( S ) is a measure of the disorder in a system. In a closed system, entropy always increases over time. … If entropy (disorder) increases, and the reaction enthalpy is exothermic ( ΔH<0 ) or weakly endothermic ( ΔH>0 & small), the reaction is generally spontaneous.
What is spontaneity of a process?
In thermodynamics, a spontaneous process is a process which occurs without any external input to the system. … Because spontaneous processes are characterized by a decrease in the system’s free energy, they do not need to be driven by an outside source of energy.
What is meant by spontaneity of reaction?
A spontaneous reaction is a reaction that favors the formation of products at the conditions under which the reaction is occurring. … This combination of a decrease in energy and an increase in entropy means that combustion reactions occur spontaneously.
How entropy affects the spontaneity of the process?
It’s the other way around: entropy affects whether a process is spontaneous. … As entropy increases, the process becomes more spontaneous. As entropy decreases, the process becomes less spontaneous.What is meant by entropy?
entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.
What is the difference between spontaneity and disorder?
The second law of thermodynamics describes the relationship between entropy and the spontaneity of natural processes. Second Law: In an isolated system, natural processes are spontaneous when they lead to an increase in disorder, or entropy.
What are the conditions for spontaneity?
Two conditions for a reaction to be spontaneous are: achieve maximum energy and achieve minimum energy. In thermodynamics, a spontaneous is the time evaluation. Processes which involve an entropy of the system are spontaneous.
How do enthalpy and entropy determine spontaneity?
Spontaneous reactions release free energy, which can be used to do work. A mathematical combination of enthalpy change and entropy change allows the change in free energy to be calculated. A reaction with a negative value for ΔG releases free energy and is thus spontaneous.What is another word for spontaneity?
In this page you can discover 30 synonyms, antonyms, idiomatic expressions, and related words for spontaneity, like: casualness, inspiration, easiness, unpremeditation, ease, tendency, impulse, informality, naturalness, spontaneousness and unceremoniousness.
What is meant by Clausius inequality?The Clausius Inequality applies to any real engine cycle and implies a negative change in entropy on the cycle. That is, the entropy given to the environment during the cycle is larger than the entropy transferred to the engine by heat from the hot reservoir.
Article first time published onWhat is entropy spontaneity Class 11?
Entropy is a measure of randomness or disorder of the system. … Entropy is a state function. The change in its value during a process, is called the entropy change. ΔS = S2 -S1 = ∑S products – ∑S reactants. 1) When a system absorbs heat ,the molecules start moving faster because kinetic energy increases.
How do entropy change occur?
Entropy increases when a substance is broken up into multiple parts. The process of dissolving increases entropy because the solute particles become separated from one another when a solution is formed. Entropy increases as temperature increases.
How does entropy apply to life?
Entropy is simply a measure of disorder and affects all aspects of our daily lives. In fact, you can think of it as nature’s tax. Left unchecked disorder increases over time. Energy disperses, and systems dissolve into chaos.
Is entropy and enthalpy the same?
Difference Between Enthalpy and EntropyEnthalpy is a kind of energyEntropy is a propertyIt is the sum of internal energy and flows energyIt is the measurement of the randomness of moleculesIt is denoted by symbol HIt is denoted by symbol S
What is entropy dummies?
Entropy is a measure of how much the atoms in a substance are free to spread out, move around, and arrange themselves in random ways. … So, when there is higher entropy, there is greater freedom for atoms to move around. And the more energy we have, the more atoms move around and spread into a random arrangement.
Is spontaneous process reversible?
Spontaneous processes are irreversible. Like total energy, E, and enthalpy, H, entropy is a state function. The second law of thermodynamics states that the entropy of the universe increases for spontaneous processes, and the entropy of the universe does not change for reversible processes.
How does spontaneity apply to a chemical reaction?
A spontaneous reaction is a reaction that favors the formation of products at the conditions under which the reaction is occurring. … This combination of a decrease in energy and an increase in entropy means that combustion reactions occur spontaneously.
What are the criterion for predicting the spontaneity of a process in terms of entropy change?
We can assess the spontaneity of the process by calculating the entropy change of the universe. If ΔSuniv is positive, then the process is spontaneous. At both temperatures, ΔSsys = 22.1 J/K and qsurr = −6.00 kJ.
What are examples of entropy?
A campfire is an example of entropy. The solid wood burns and becomes ash, smoke and gases, all of which spread energy outwards more easily than the solid fuel. Ice melting, salt or sugar dissolving, making popcorn and boiling water for tea are processes with increasing entropy in your kitchen.
What does the law of entropy say?
The second law of thermodynamics can be stated in terms of entropy. If a reversible process occurs, there is no net change in entropy. In an irreversible process, entropy always increases, so the change in entropy is positive. The total entropy of the universe is continually increasing.
What does it mean if entropy is negative?
Entropy is the amount of disorder in a system. Negative entropy means that something is becoming less disordered. In order for something to become less disordered, energy must be used. This will not occur spontaneously.
Why is spontaneity important in a relationship?
It can mean something way more everyday, but possibly way more powerful for a great relationship. ‘ … “Couples and people are constantly changing, so keeping spontaneity alive in a relationship helps both people grow with each other through new-found interests and reduces the risk of growing apart.”
Is spontaneity a real word?
noun, plural spon·ta·ne·i·ties. the state, quality, or fact of being spontaneous. spontaneous activity.
Is Unspontaneous a word?
Not spontaneous; not voluntary; forced; artificial: as, unspontaneous laughter.
Are Spertaneous reactions Exergonic?
Exergonic reactions are also called spontaneous reactions, because they can occur without the addition of energy. Reactions with a positive ∆G (∆G > 0), on the other hand, require an input of energy and are called endergonic reactions.
How is enthalpy related to the spontaneity of a reaction?
High temperatures make the reaction more favorable. When ΔS is negative and ΔH is negative, the relative magnitudes of ΔS and ΔH determine if the reaction is spontaneous. Low temperatures make the reaction more favorable. When ΔS is negative and ΔH is positive, a process is not spontaneous at any temperature.
What is Kelvin Planck and Clausius statement?
Therefore, the heat transfer is an irreversible process that only occurs in one direction. The Kelvin-Planck statement is the statement of the second law of thermodynamics for heat engines, while the Clausius statement is the statement of the refrigerators and heat pumps.
Does entropy ever decrease?
The total entropy of a system either increases or remains constant in any process; it never decreases. For example, heat transfer cannot occur spontaneously from cold to hot, because entropy would decrease.
What is Unattainability of absolute zero?
On the one hand, we have the Principle of Unattainability of absolute zero which states that cooling any system to absolute zero temperature in a finite number of steps and within a finite time is physically impossible by any procedure, no matter how idealised the procedure.
What is entropy Shaalaa?
Definition of entropy: The property of a system which measures the degree of disorder or randomness in the system is called entropy. It is denoted by letter S. Concept: Chemical Thermodynamics and Energetic – Free Energy Change for Spontaneous and Non Spontaneous Processes.
What is entropy Mcq?
Entropy is a measure of randomness or disorder in the system. Entropy is a thermodynamic function and is denoted by S. The higher the entropy more the disorder in the isolated system. A change in entropy in a chemical reaction is related to the rearrangement of atoms from reactants to products.