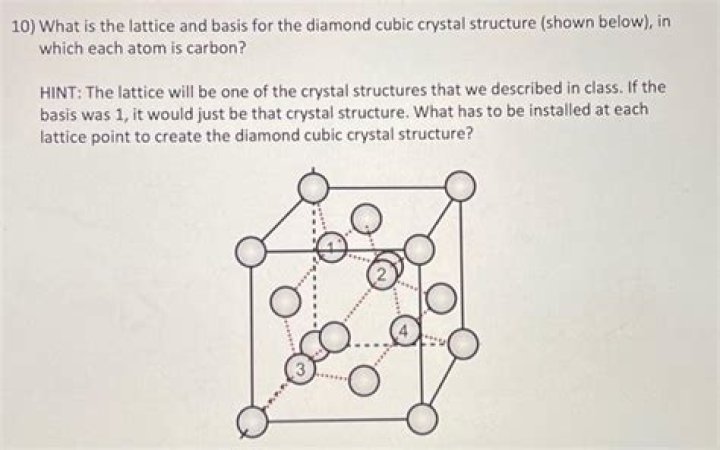
Diamond is a crystal structure with a face centered cubic Bravais lattice and two atoms in the basis. Carbon, silicon germanium, and α-tin form this crystal structure.
What type of lattice is diamond?
The diamond lattice can be viewed as a pair of intersecting face-centered cubic lattices, with each separated by 14 of the width of the unit cell in each dimension.
What is lattice structure of solid diamond?
Diamond is a crystal structure with a face centred cubic space lattice and two identical atoms in the basis. One atom sits on the lattice point whereas the other one is shifted by 14 along each axis. This forms a tetrahedral structure where each atom is surrounded by four equal-distanced neighbours.
What is lattice and basis in crystal structure?
A lattice is a hypothetical regular and periodic arrangement of points in space. It is used to describe the structure of a crystal. … A basis is a collection of atoms in particular fixed arrangement in space.Is diamond cubic structure FCC?
1.5. 1 Diamond Cubic Structure For example, in diamond, the base lattice is FCC and is built by the C atoms with half of the tetrahedral sites filled by C atoms. Thus, the unit cell of diamond contains a total of 8 atoms. The structure is typically called as diamond cubic structure.
What is cubic diamond?
Cubic zirconia is a much less expensive man-made gem that appears quite like a diamond, but they are very different. … Synthetic diamonds are also made in labs, but they have the same carbon structure as diamonds- cubic zirconias do not.
What is the basis of diamond?
Diamond is a crystal structure with a face centered cubic Bravais lattice and two atoms in the basis. Carbon, silicon germanium, and α-tin form this crystal structure.
What is basis in crystal physics?
The basis is the arrangement of atoms associated with each lattice point. Sometimes there is only one atom per lattice point – a monatomic lattice – but often there are more. Mathematically, this association of one copy of something with every point is a convolution.What is a basis crystal?
The crystal basis is the arrangement of atoms that is particular to the mineral being considered. … The unit cells are repeated over and over again in three dimensions to make up a macroscopic crystal. The repeating units can be chosen to have different sizes, but the minimum size possible is the primitive unit cell.
What are the lattice parameters of cubic crystal system?This crystal system has the lowest symmetry and must be described by 3 lattice parameters (a, b, and c) and the 3 angles (α, β, and γ).
Article first time published onHow is diamond crystal structure formed?
Diamond crystal is constituted by sp3 hybridized carbon atoms which are bonded to four nearest neighbors in tetrahedral coordination. When the crystal is cut or cleaved, bonds are broken, creating dangling bonds at the surface. These dangling bonds are the source of chemical activity on diamond surfaces.
Is diamond cubic or tetrahedral?
In a diamond, the carbon atoms are arranged tetrahedrally. Each carbon atom is attached to four other carbon atoms 1.544 x 10-10 meter away with a C-C-C bond angle of 109.5 degrees. It is a strong, rigid three-dimensional structure that results in an infinite network of atoms.
How do you find the lattice parameter of a diamond?
The diamond lattice is face-centered cubic. The simplified packing fraction is 8 x (V atom) / V unit cell. After making substitutions for known volume of spheres and cubes and simplifying, the equation becomes √3 x π/16 with a solution of 0.3401.
What is the packing factor of diamond cubic crystal structure?
Answer. Packing factor of diamond cubic structure is 0.34. The equation for finding the packing fraction is No of atoms in unit cell ×Volume of atom/Volume of unit cell. Diamond has eight atoms per unit cell.
What will be the density of diamond structure?
10.12 g/cm3.
What is the crystal shape of diamond?
The most common shape for rough gem quality diamond is the octahedron, which looks like two pyramids back to back. Crystals that are almost perfect in shape and transparency are called glassies.
Is cubic zirconia crystal?
Cubic zirconia (CZ) is the cubic crystalline form of zirconium dioxide (ZrO2). The synthesized material is hard and usually colorless, but may be made in a variety of different colors. It should not be confused with zircon, which is a zirconium silicate (ZrSiO4). It is sometimes erroneously called cubic zirconium.
What are cubic zirconia stones?
Cubic Zirconia (CZ) is an inexpensive diamond alternative with many of the same qualities as a diamond. This crystalline material (or CZ) is synthetic, which means it is created in a laboratory.
Is diamond cubic structure close packed?
The diamond structure is a very common form. This structure is based on the cubic close packed structure with 4 additional atoms (pictured as green balls) in holes within the structure. The form of carbon in diamonds has this structure. It is also the structure of crystalline silicon.
What is called crystal lattice?
Crystal lattice is a regular arrangement of the constituent atoms or ions or molecules in three-dimensional space. A crystal lattice also called a space lattice or simply, a lattice. There are only 14 possible three-dimensional lattices. These are called Bravais’ lattices.
How does crystal differ from lattice?
What is the difference between Lattice and Crystal? Lattice describes the structure of crystals. When a group of molecules tends to arrange each unit repeatedly at a lattice point, a crystal is made. In a crystal structure, there is a pattern of arranging the atoms or units.
What is the lattice point?
A lattice point is a point at the intersection of two or more grid lines in a regularly spaced array of points, which is a point lattice. In a plane, point lattices can be constructed having unit cells in the shape of a square, rectangle, hexagon, and other shapes.
What is basis vector in crystal structure?
Basis vectors specify how the various entities that make up the basis, are connected. Lattice vectors then define the primitive cell (in terms of the basis unit) and hence the whole crystal.
What is lattice in chemistry?
A lattice is an ordered array of points describing the arrangement of particles that form a crystal. The unit cell of a crystal is defined by the lattice points. … In the structure drawn, all of the particles (yellow) are the same.
What is the reciprocal lattice to simple cubic lattice?
The reciprocal lattice of the simple cubic lattice is itself a simple cubic lattice with the length of each side being 2π/a. Show that the reciprocal lattice of the fcc lattice is the bcc lattice.
What is the structure of cubic?
The simple cubic structure (sc) In the simple cubic structure there is only one lattice point at each corner of the cube-shaped unit cell. They mark the position of either a single atom, or the same group of atoms, known as the motif, which is repeated across the lattice.
How do you find the lattice parameter of a cubic equation?
Calculate the lattice constant, a, of the cubic unit cell. If the space lattice is SC, the lattice constant is given by the formula a = [2 x r]. For example, the lattice constant of the SC-crystallized polonium is [2 x 0.167 nm], or 0.334 nm.
What are the different space lattice in the cubic system?
There are three main varieties of these crystals: Primitive cubic (abbreviated cP and alternatively called simple cubic) Body-centered cubic (abbreviated cI or bcc) Face-centered cubic (abbreviated cF or fcc, and alternatively called cubic close-packed or ccp)
What is the coordination number of diamond cubic crystal structure?
In diamond, coordination number of carbon atoms is 4 and its unit cell has 8 carbon atoms.
Why is a diamond a structure?
All the carbon atoms of Diamond are said to possess strong chemical bonds with that of the four other carbon atoms, thus making a perfect tetrahedron structure and on throughout the crystal. The carbon atoms, here are sp3 hybridized, and the bond lengths of carbon-carbon atom are equal.
What is lattice parameter of BCC?
In BCC iron, carbon atoms enter tetrahedral sites, such as 1/4, 1/2, 0. The lattice parameter is 0.3571 nm for FCC iron and 0.2866 nm for BCC iron.