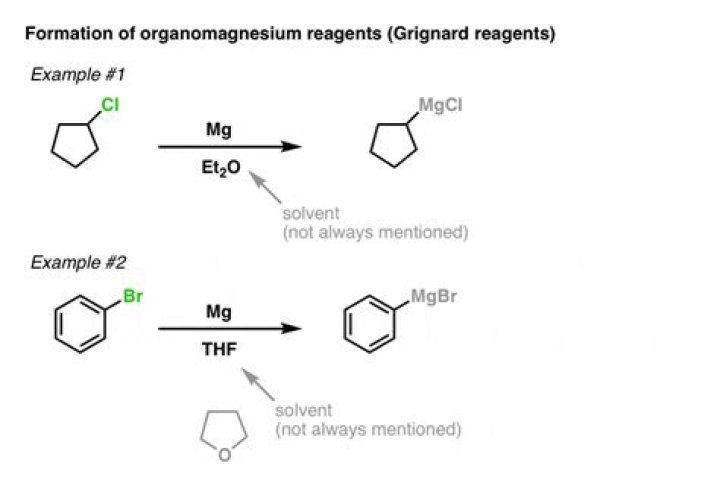
Explanation: A Grignard reaction involves the reaction of an alkyl (or aryl halide) with magnesium metal to form an alkylmagnesium halide. Diethyl ether is an especially good solvent for the formation of Grignard reagents
What happens when Haloalkanes reacts with magnesium in presence of dry ether?
Haloalkanes and aryl and vinyl halides react with magnesium metal to yield organomagnesium halides called Grignard reagents. An ether solvent, usually diethyl ether, is required for preparation of Grignard reagents.
What does diethyl ether do in Grignard reaction?
q Diethyl ether is an especially good solvent for the formation of Grignard reagents because ethers are non-acidic (aprotic). Water or alcohols would protonate and thus destroy the Grignard reagent, because the Grignard carbon is highly nucleophilic. This would form a hydrocarbon.
What type of reaction is a Grignard reaction?
The Grignard reaction (French: [ɡʁiɲaʁ]) is an organometallic chemical reaction in which alkyl, allyl, vinyl, or aryl-magnesium halides (Grignard reagent) is added to a carbonyl group in an aldehyde or ketone. This reaction is important for the formation of carbon–carbon bonds.What happens when alkyl halide react with magnesium in presence of ether?
magnesium halide. Grignard’s reagent. alkene.
Which Alkylhalide quickly gives Grignard reagent when reacts with magnesium metal in dry ether?
C2H5OH.
Which of the following can give Grignard reagent on reacting with magnesium in anhydrous ether?
C2H5Cl can give Grignard reagent when reacted with Mg in dry ether.
What happens when ester reacts with Grignard reagent?
An ester (or an acid halide or an anhydride) reacts first with a Grignard reagent to form a ketone, which reacts further to give an alcohol. … An organocadmium compound, for instance, formed from a Grignard reagent by reaction with cadmium chloride, yields a ketone when treated with an acid chloride.Why magnesium is used in Grignard reagent?
Magnesium. Typically the reaction to form Grignard reagents involves the use of magnesium ribbon. All magnesium is coated with a passivating layer of magnesium oxide, which inhibits reactions with the organic halide.
What is the action of alkyl halide on magnesium?However, both alkyl and aryl halides react with magnesium to form Grignard reagents. The alkali metals (Li, Na, K etc.) and the alkaline earth metals (Mg and Ca, together with Zn) are good reducing agents, the former being stronger than the latter. These same metals reduce the carbon-halogen bonds of alkyl halides.
Article first time published onIs magnesium soluble in ether?
Ether is used as a solvent because it is aprotic and can solvate the magnesium ion.
Why must the ether be anhydrous?
For a variety of reasons, anhydrous diethyl ether is the solvent of choice for carrying out a Grignard synthesis. Vapors from the highly volatile solvent help to prevent oxygen from reaching the reaction solution. … Once the reaction begins, it will continue to reflux in the absence of an external heat source.
Why are ether solvents used in the preparation of Grignard and organolithium reagents?
Ethyl ether or THF are essential for Grignard reagent formation. Lone pair electrons from two ether molecules form a complex with the magnesium in the Grignard reagent (As pictured below). This complex helps stabilize the organometallic and increases its ability to react.
Which of the following compounds react with magnesium yield Grignard reagent?
Complete answer: Methyl magnesium iodide is a Grignard reagent. Grignard reagents are highly basic in nature, so they can react with any species that donates a proton.
Which of the reactions are most common in alkyl halides?
Alkyl halides can undergo two major types of reactions – substitution and/or elimination. The substitution reaction is called a Nucleophilic Substitution reaction because the electrophilic alkyl halide forms a new bond with the nucleophile which substitutes for (replaces) the halogen at the alpha-carbon.
Which of the following on reaction with Grignards reagent produces alkane?
We know that Grignard reagent produces alkane if react with (1)phenol because, as per hint phenol give it’s H+ easily then other respective group mentioned as well as carboxylic group is a strong acid where Grignard’s reagent is a strong base therefore, they form salt and water when react also (3)amine can produce …
What happens when sodium ethoxide react with chloromethane?
When sodium ethoxide is reacted with ethanoyl chloride it results in the formation of an ester compound that is ethyl ethanoate. In this reaction, C2H5O− ,this is a strong nucleophile attacks on a carbonyl complex having slightly positive charge on carbon and a slightly negative charge is formed on oxygen.
How does chlorobenzene react with magnesium?
Firstly, we convert to chlorobenzene to phenyl magnesium chloride. In step-1, the reaction takes place when chlorobenzene is reacted with magnesium in the presence of dry ether then it results in the formation of phenyl magnesium chloride which is also known as Grignard’s reagent.
What happens when ethyl bromide reacts with sodium metal?
Butane is formed when ethyl bromide is treated with sodium metal in dry ether. When sodium metals are reacted with alkyl bromides in the presence of dry ether, alkanes are produced. This reaction is called Wurtz reaction.
When alkyl halide is treated with magnesium in dry ether as solvent It gives?
An alkyl halide (A), on reaction with magnesium in dry ether followed by treatment with ethanol gave 2-methylbutane.
Which reagent is used in the conversion of alkyl halide to Ester?
The reagent used to convert alkyl halide to ester is silver carboxylate (RCOOAg).
How is ch4 prepared by Grignard reagent?
Methyl magnesium bromide reacts with ethanol to give methane. Methyl group in the grignard reagent has a lone pair with a negatice charge. This methyl group attacks the hydrogen atom of -OH group and takes that hydrogen atom towards methyl group. It forms methane.
Is magnesium a halide?
Magnesium in its 2+ oxidation state forms salts with a variety of counterions. All the four magnesium halides are known and are commercially available. Of these, magnesium bromide and iodide are often used as Lewis acids in a variety of stereoselective transformations.
How does iodine activate magnesium?
Various methods of magnesium surface activation are tested. For example, iodine and ferric chloride increase reactivity by increasing the density of reactive sites and by increasing the rate at which individual sites react.
Why Grignard reagent is prepared in anhydrous condition?
Grignard reagent should be prepared under anhydrous conditions, because it is very reactive. It reacts very quickly with any source of proton to give hydrocarbon. It reacts with water very quickly. Therefore, it is necessary to avoid moisture from the Grignard reagents.
What is an ester reaction?
Esterification is the general name for a chemical reaction in which two reactants (typically an alcohol and an acid) form an ester as the reaction product. Esters are common in organic chemistry and biological materials, and often have a pleasant characteristic, fruity odor.
Which compound can be prepared from the reaction of an ester and a Grignard reagent?
ethyl acetate). Esters are still reactive enough to undergo hydrolysis to form carboxylic acids, alcoholysis, to form different esters, and aminolysis to form amides. Also, they can react with Grignard reagents to form 3o alcohols and hydride reagents to form 1o alcohols or aldehydes.
What do Grignards react with?
Grignard reagents are formed by the reaction of magnesium metal with alkyl or alkenyl halides. They’re extremely good nucleophiles, reacting with electrophiles such as carbonyl compounds (aldehydes, ketones, esters, carbon dioxide, etc) and epoxides.
When phenyl magnesium bromide reacts with T butanol the product would be?
When phenyl magnesium bromide reacts with t-butanol, the product would be. Phenyl magnesium bromide reacts with t-butanol to produce benzene because phenyl (electronegative group) is associated with active hydrogen of alcohol i.e. – H of -OH group of alcohol.
Which compound gives a primary alcohol by reacting with alkyl magnesium bromide followed by hydrolysis?
Ketones on reaction with Grignard reagent (alkyl magnesium halide) followed by hydrolysis will always yield tertiary alcohol (3∘ alcohol) while aldehydes except formaldehyde (HCHO H C H O ) gives secondary alcohol and formaldehyde gives primary alcohol.
What is the order of reactivity of alkyl halides towards elimination reaction?
Answer: The reactivity of the alkyl halide is decided by the ease with which the halide leaves the substrate. As per the leaving ability, the order is I>Br>Cl>F.