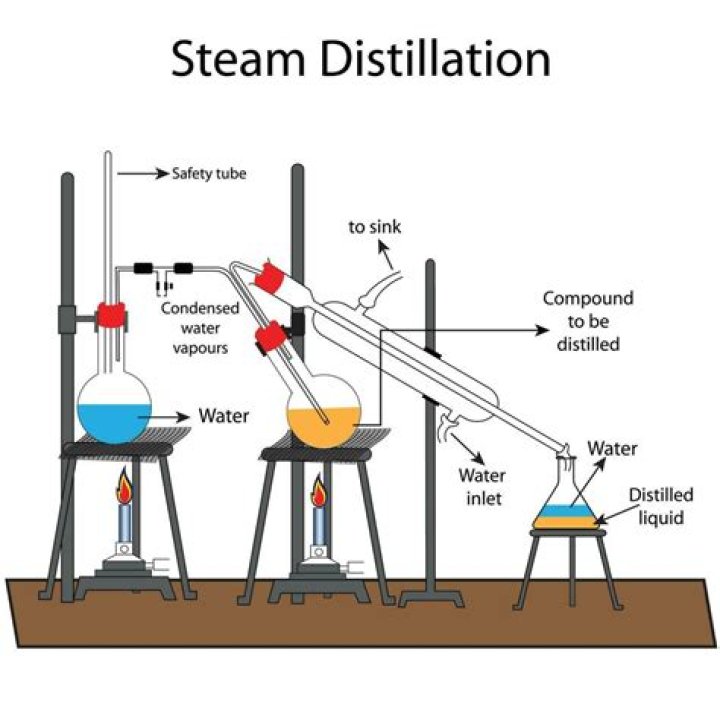
Essential oils are purified by steam distillation.
What is example of steam distillation?
Steam distillation is often employed in the isolation of essential oils, for use in perfumes, for example. In this method, steam is passed through the plant material containing the desired oils. Eucalyptus oil, camphor oil and orange oil are obtained by this method on an industrial scale.
Which of the following can be purified by distillation?
Distillation method is used for refining low boiling metals like Zinc and Mercury.
Which can be separated by steam distillation?
Ortho and para-nitrophenols can be separated by steam distillation.What is produced by steam distillation?
Steam distillation of biomass generally yields two products: a relatively high purity essential oil and an aqueous condensate called a hydrosol. The oil consists of hydrophobic, often aromatic compounds that are produced in very small concentrations as secondary metabolites in the plants.
What type of organic compounds can be purified by distillation?
The method is commonly used for those liquids which are sufficiently stable at their boiling points and which contain non-volatile impurities. For example: simple organic liquids such as benzene, ethanol, acetone, chloroform, carbon tetrachloride, toluene, xylenes, etc. can be purified by simple distillation.
What is steam distillation Class 8?
Steam distillation is a separation process which purifies isolate temperature-sensitive materials, such as natural aromatic compounds. … Steam distillation implements low-pressure steam, this replaces the volatile compounds from the intact plant material.
What are the 5 example of distillation?
Examples of Distillation Salt water is turned into fresh water through distillation. Various forms of fuel, such as gasoline, are separated from crude oil by distillation. Alcoholic beverages are made through distillation. The alcohol is boiled off from the rest of the mixture and collected in a concentrated format.Why steam is used in steam distillation?
Steam or water is added to the distillation apparatus, lowering the boiling points of the compounds. The goal is to heat and separate the components at temperatures below their decomposition point.
Why is steam distillation The preferred method for extracting limonene?Steam distillation is a much gentler way to obtain essential oils, as it allows the distillation of co-mixture of oil and water at a boiling point less than 100 °C. Limonene is responsible for the characteristic smell of citrus peel.
Article first time published onCan Acetone be purified by steam distillation?
The process involves the heating of the mixture so that the component with low boiling point escapes first followed by the other. The acetone and methanol are volatile liquids. The boiling point of liquids is close to each other. Thus, a suitable and sophisticated distillation technique is used to separate the liquids.
Which substance can be purified by crystallization?
Potash alum, tetraamine copper sulphate, washing soda are purified by crystallization. Explanation: Crystallisation is a very popular technique to separate a solute from a solvent.
Can P Hydroxybenzaldehyde be purified by steam distillation?
P – hydroxybenzaldehyde has inter molecular hydrogen bond so not purified by steam distillation.
Who founded steam distillation?
Petroleum was first distilled by another Muslim chemist al-Razi (Rhazes) in the 9th century, for producing kerosene, while steam distillation was invented by Avicenna in the early 11th century, for producing essential oils.
What is Hydrodistillation process?
Hydrodistillation is a traditional method for the extraction of bioactive compounds from plants. In this method, plant materials are packed in a still compartment then water is added in sufficient amount and brought to a boil. Alternatively, direct steam may also be injected into the plant sample.
How is liquid purified by distillation process?
Distillation is used to purify a compound by separating it from a non-volatile or less-volatile material. When different compounds in a mixture have different boiling points, they separate into individual components when the mixture is carefully distilled.
What are the types of purification?
- Simple crystallisation.
- Fractional crystallisation.
- Sublimation.
- Simple distillation.
- Fractional distillation.
- Distillation under reduced pressure.
- Steam distillation.
- Azeotropic distillation.
How organic compounds are purified?
Distillation is used to identify and purify organic compounds. In the process of purification, we separate a compound from another material by exploiting their boiling points. When different compounds in a mixture have different boiling points, the mixture separates into its component parts when it is distilled.
Why is steam distillation used for eugenol?
Eugenol has a high boiling point (254 oC), and many organic compounds decompose at such high temperatures. Steam distillation allows eugenol to be distilled at a much lower boiling point (< 100 oC), thus minimizing the potential for decomposition.
How is steam distillation different from simple distillation?
Different. Simple distillation is powered by a direct heat source usually of over 100°C. In steam distillation the heat source is steam passing through the medium being distilled. It is therefore usually performed at much lower temperature except where the steam is under pressure.
How is alcohol purified?
Ethanol is purified almost only by distillation in the industry. Although distillation is one of the most effective liquid-liquid separation techniques, it contains some critical disadvantage, cost and limitation on separation of volatile organic compounds.
What is a distillation example?
Distillation is the process of separating components of a mixture based on different boiling points. Examples of uses of distillation include purification of alcohol, desalination, crude oil refining, and making liquefied gases from air. Humans have been using distillation since at least 3000 BC in the Indus valley.
Why is an ice bath used in distillation?
Since the vapor pressure of water at 20 °C is roughly 2.3 kPa, then 2.3/101.325 or 0.023 would be the fraction of water that would not condense and would pass out of the condenser. This is why the distillate is frequently chilled in an ice bath during the distillation.
Why is CO2 extraction better than steam distillation?
In steam distillation, the molecular composition of both the plant matter and the essential oil are changed due to the temperature applied. On the other hand, a CO2 extract is closer in chemical composition to the original plant from which it is derived, as it contains a wider range of the plant’s constituents.
What is the name of the extraction method used to extract lemongrass and spices?
It is also used to produce perfumes, avours, detergents, and pharmaceuticals. The method found to be the most suitable for the extraction of lemongrass essential oil is steam distillation, since it allows obtaining the oil without altering product quality. The chemical composition of the essential oil of C.
What is the difference between Hydrodistillation and steam distillation?
The key difference between steam distillation and hydrodistillation is that steam distillation uses steam for the extraction whereas hydrodistillation uses water, steam or a combination of water and steam for the extraction.
Is aniline purified by steam distillation?
Aniline is purified using the process of steam distillation because aniline is steam volatile and it is insoluble in water.
Which is purified by steam distillation aniline benzoic acid?
Explanation: Aniline is insoluble in water and possess low vapour pressure. Hence it can be purified by steam distillation.
How camphor can be purified?
Camphor can easily be purified by the process of sublimation. Sublimation is used for separating substances with on heating change directly into vapour. Camphor gets directly converted to its gaseous state, leaving the salt behind. Camphor is separated from NaCl by Sublimation.
How is common salt purified?
The common salt is purified by the process of crystallization.
How does crystallisation purify?
Crystallization is used in the chemistry laboratory as a purification technique for solids. An impure solid is completely dissolved in a minimal amount of hot, boiling solvent, and the hot solution is allowed to slowly cool. … The crystallized solid is then filtered away from the impurities.