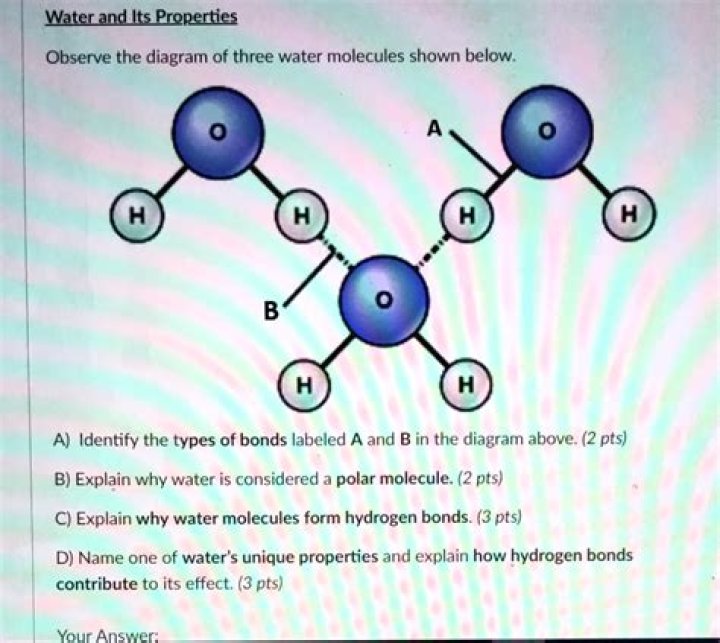
Water is a chemical compound and polar molecule, which is liquid at standard temperature and pressure. It has the chemical formula H2O, meaning that one molecule of water is composed of two hydrogen atoms and one oxygen atom. Water is found almost everywhere on earth and is required by all known life.
Which is correct in a water molecule?
Detailed Solution. Option 3 is Correct. The water molecule has a bent-shaped structure. In water, two hydrogen atoms are attached to one central atom of oxygen by covalent bond, making an angular spatial structure.
What is true about water molecule bonding?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). … The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom.
Are water molecules polar or nonpolar?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).What do all water molecules have?
A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
What is the structure of water molecules?
What is the structure of a water molecule? A water molecule consists of two hydrogen atoms and one oxygen atom. The three atoms make an angle; the H-O-H angle is approximately 104.5 degrees. The center of each hydrogen atom is approximately 0.0957 nm from the center of the oxygen atom.
How do water molecules form droplets of water?
It turns out that this surface tension is the result of the tendency of water molecules to attract one another (called cohesion). … When cohesion is more of a factor, the water forms spherical droplets; when adhesion is more of a factor, we get sheets of water.
What is it called when water molecules are attracted to other water molecules?
The property of cohesion describes the ability of water molecules to be attracted to other water molecules, which allows water to be a “sticky” liquid. … The attraction between individual water molecules creates a bond known as a hydrogen bond.Why do we describe a water molecule as polar?
Since oxygen has a higher electronegativity than hydrogen, the electrons of the molecule tend to group closer to the oxygen than to the hydrogen atoms. … Therefore, water is said to be a “polar” molecule, which means that there is an uneven distribution of electron density.
Which statement best describes why a water molecule is polar?Water is a polar molecule because the bonds between oxygen and hydrogen are polar covalent bonds. This means that although they form covalent bonds (normally non-polar) the difference in electronegativity is great enough to make one side more negative (oxygen side) and the other side more positive (hydrogen side).
Article first time published onWhy do water molecules stick to other water molecules quizlet?
Water molecules stick to other water molecules. This property is called cohesion. … Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts the negatively charged oxygen end of another water molecule.
What type of bond is H2O?
H2O is a Covalent bond, as the two elements are non-metals. A water molecule is a simple molecule, and simple molecules consist of a small number of atoms joined by covalent bonds.
What properties make water such a special molecule?
Water molecules are polar, so they form hydrogen bonds. This gives water unique properties, such as a relatively high boiling point, high specific heat, cohesion, adhesion and density.
Is a water molecule water?
From a chemical perspective, water is a molecule in which a single oxygen atom is linked to two hydrogen atoms. It is less well known that water exists in two different forms (isomers) at the molecular level. The difference lies in the relative orientation of the nuclear spins of the two hydrogen atoms.
How do water molecules work?
Water molecules are naturally attracted and stick to each other because of this polarity, forming a hydrogen bond. This hydrogen bond is the reason behind many of water’s special properties, such as the fact that it’s denser in its liquid state than in its solid state (ice floats on water).
What are Waters properties?
- Water is polar. …
- Water is an excellent solvent. …
- Water has high heat capacity. …
- Water has high heat of vaporization. …
- Water has cohesive and adhesive properties. …
- Water is less dense as a solid than as a liquid.
What is it about water molecules that helps water keeps its shape the moment the balloon is popped?
Why do you think the water keeps its shape the moment the balloon is popped? Students should realize that water holds together pretty well because the water molecules are attracted to each other.
How does the structure of the water molecule help explain the ability of water to form beads on the wax?
Water forms tiny beads on top of the wax instead of soaking through it. … The polarity allows multiple hydrogen bonds to form among adjacent water molecules. The hydrogen bonds attract water molecules to one another, and account for the ability of water molecules to adhere, or stick together, such as by forming beads.
Why do hydrophobic molecules repel water?
Hydrophobic molecules and surfaces repel water. … Without opposite electrical charges on the molecules, water cannot form hydrogen bonds with the molecules. The water molecules then form more hydrogen bonds with themselves and the nonpolar molecules clump together.
What is the shape of water molecules?
The molecular geometry of the water molecule is bent. The H-O-H bond angle is 104.5°, which is smaller than the bond angle in NH3 (see Figure 11).
Why does the water molecule have polarity quizlet?
A water molecule is polar because there is an uneven distribution of electrons between the oxygen and hydrogen atoms. … Each molecule of water can form multiple hydrogen bonds with other water molecules.
Why is water polar quizlet?
chemical bond in which atoms share one pair of electrons. … water is called polar molecule, its two hydrogen atoms are joined to one oxygen atom by single covalent bonds, but the electrons of the covalent bonds are not shared equally between oxygen and hydrogen this unequal sharing makes water a polar molecule.
Why do water molecules attract each other?
The two hydrogen atoms and one oxygen atom within water molecules (H2O) form polar covalent bonds. … As a result of water’s polarity, each water molecule attracts other water molecules because of the opposite charges between them, forming hydrogen bonds.
What is the attraction between water molecules and other molecules allowing water to climb up a straw?
These cohesive forces are also related to the water’s property of adhesion, or the attraction between water molecules and other molecules. This is observed when water “climbs” up a straw placed in a glass of water. You will notice that the water appears to be higher on the sides of the straw than in the middle.
Why is the arrangement of water molecules is different in ice and water?
Ice has a very regular pattern with the molecules rigidly apart from one another connected by the hydrogen bonds that form a crystalline lattice. These crystals have a number of open regions and pockets making ice less dense than liquid water. This is why ice floats on water.
Which statement correctly describes the electrons in a water molecule?
Which statement correctly describes the electrons in a water molecule? Electrons are pulled closer to the oxygen atom. How does a catalyst influence a chemical reaction?
Why are the bonds in a water molecule polar covalent and how does that affect the interactions between water molecules?
The two hydrogen atoms and one oxygen atom within water molecules (H2O) form polar covalent bonds. … As a result of water’s polarity, each water molecule attracts other water molecules because of the opposite charges between them, forming hydrogen bonds.
Which best describes what a polar molecule is?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A diatomic molecule that consists of a polar covalent bond, such as HF, is a polar molecule. … A molecule with two poles is called a dipole (see Figure below ) . Hydrogen fluoride is a dipole.
Is the tendency of water molecules to stick together?
The tendency for water molecules to form weak bonds and stick to each other is called cohesion. Because of cohesion, water molecules remain joined together as they move within or between the cells of organisms.
Which statement is true about the electrons in each polar bond of a water molecule?
Which statement is true about the electrons in each polar bond of a water molecule? They are more attracted by the oxygen atom than by the hydrogen atom. Which best explains why water has a high specific heat?
Is h20 ionic or molecular?
H2O is a covalent molecule. It’s also polar molecule. polar molecule occur when there is electronegativity difference (0.5 to 2.0) between bonded atoms. The Hydrogen and Oxygen respective electronegatives of 3.44 and 2.20.