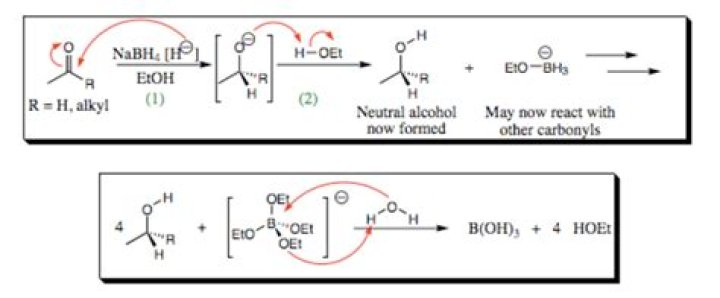
At this point, the reactivity is too weak to occur without a stronger reducing agent. The short answer is that NaBH4 is simply too weak a reducing agent for carboxylic acids and esters and the two will simply mingle in solution with little to no reaction for months.
Does NaBH4 affect esters?
What it’s used for: Sodium borohydride is a good reducing agent. … By itself, it will generally not reduce esters, carboxylic acids, or amides (although it will reduce acyl chlorides to alcohols).
Can esters get reduced?
Esters can be reduced to 1° alcohols using LiAlH4 Esters can be converted to 1o alcohols using LiAlH4, while sodium borohydride (NaBH4) is not a strong enough reducing agent to perform this reaction.
Which is not reduced by NaBH4?
NaBH4 is effective reducing agent for the reduction of aldehydes, ketones and acyl chloride to alcohol but it cannot reduce esters, carboxylic acids and amides.Why is NaBH4 preferred over LiAlH4?
The key difference between LiAlH4 and NaBH4 is that LiAlH4 can reduce esters, amides and carboxylic acids whereas NaBH4 cannot reduce them. … But LiAlH4 is a very strong reducing agent than NaBH4 because the Al-H bond in the LiAlH4 is weaker than the B-H bond in NaBH4. This makes the Al-H bond less stable.
Can sodium borohydride reduce ethers?
Sodium borohydride is relatively insoluble in ether solvents, so these are rarely used for borohydride reductions. … Ethanolic solutions of sodium borohydride reduce aldehydes and ketones in the presence of epoxides, esters, lactones, acids, nitriles, or nitro groups.
Why NaBH4 Cannot reduce primary halides?
In NaBH4, the negative charge is developed by the non metal, B, which is relatively stable. This slows down the reaction making the hydride a weaker nucleophile. This favours SN1, which does not occur for 1°.
Can NaBH4 reduce imine?
Sodium borohydride: NaBH4 Also effective for reducing imines.Why does NaBH4 not reduce carboxylic acids?
Reduction of Carboxylic Acids The carbonyl carbon of a carboxylic acid is even more electrophilic than the carbonyl carbon in an aldehyde or ketone. However, there is also an acid proton from the carboxylic acid that can react with hydride reagents. For this reason, sodium borohydride does not reduce a carboxylic acid.
Which reagent is used to partially reduce esters to aldehydes?Conversion of Esters to Aldehydes: Hydride Reduction Much like acid chlorides, esters can be converted to aldehydes using the weaker reducing reagent diisobutylaluminum hydride (DIBALH). As shown above, an aldehyde intermediate is produced after an ester undergoes nucleophilic acyl substitution with a hydride.
Article first time published onWhy NaBH4 Cannot reduce alkenes?
NaBH4 is less reactive than LiAlH4 but is otherwise similar. … It is also convenient that, although LiAlH4 is strong enough to reduce the C=C Page 2 of a conjugated carbonyl compound, NaBH4 is not; thus the carbonyl group can be reduced without the alkene.
Why is libh4 more reactive than NaBH4?
In mixtures of methanol and diethyl ether, lithium borohydride is able to reduce esters to alcohols and primary amides to amines. In contrast, these substrates are unaffected by sodium borohydride. The enhanced reactivity is attributed to the polarization of the carbonyl substrate by complexation to the lithium cation.
Does NaBH4 reduce carboxylic acids?
Standard organic chemistry texts discuss the lower re- activity of NaBH4 compared with lithium aluminum hy- dride, LiAlH4: whereas LiAlH4 reduces carboxylic acids to primary alcohols, NaBH4 does not reduce carboxylic acids.
Does NaBH4 reduce double bond?
LiAlH4 reduces double bond only when the double bond is Beta-arly , NaBH4 does not reduce double bond. if you want to you can use H2/Ni to reduce a double bond.
Can NaBH4 reduce nitro groups?
[Google Scholar] It is known that solely sodium borohydride does not reduce nitro compounds under ordinary conditions. However, the reducing power of this reagent or its polymeric analogue (BER) undergoes a drastic change toward reduction of nitro groups by the combination with metal halides or salts2.
Which of the following Cannot be reduced by LiAlH4?
* LiAlH4 can reduce aldehydes to primary alcohols, ketones to secondary alcohols, carboxylic acids and esters to primary alcohols, amides and nitriles to amines, epoxides to alcohols and lactones to diols. * Lithium aluminium hydride cannot reduce an isolated non-polar multiple bond like C=C.
Can NaBH4 reduce carbon nitrogen double bond?
NaBH4 is not able to reduce isolated C=C bond. But, NaBH4 reduces the double bond present in an enal (a conjugated aldehyde).
Does borane reduce esters?
Borane also reduces aldehydes, ketones, lactones, epoxides, acids, tertiary amides, and nitriles but does not reduce esters.
How do you reduce an ester to an aldehyde?
Esters can be converted to aldehydes using diisobutylaluminum hydride (DIBAH). The reaction is usually carried out at -78 oC to prevent reaction with the aldehyde product.
Does NaBH4 reduce alkenes?
No. An alkene is Nucleophile. Thats why it reacts with electrophiles. NaBH4 has hydride ion.
Can NaBH4 react with alkenes?
Use of unmodified sodium borohydride would result in a 1,4 conjugate addition reaction, saturating the alkene, with a subsequent reduction of the ketone to an alcohol. Use of lithium aluminum hydride would give the same product as use of unmodified sodium borohydride, following the same reduction mechanism.
Can libh4 reduce ketone?
Lithium borohydride is commonly used for the selective reduction of esters and lactones to the corresponding alcohols in the presence of carboxylic acids, tertiary amides, and nitriles. Aldehydes, ketones, epoxides, and several other functional groups can also be reduced by lithium borohydride.
Can libh4 reduce carboxylic acid?
LITHIUM BOROHYDRIDE Allows for selective reduction of esters in the presence of carboxylic acids, amides and nitriles. Also reacts with aldehydes, ketones and epoxides.