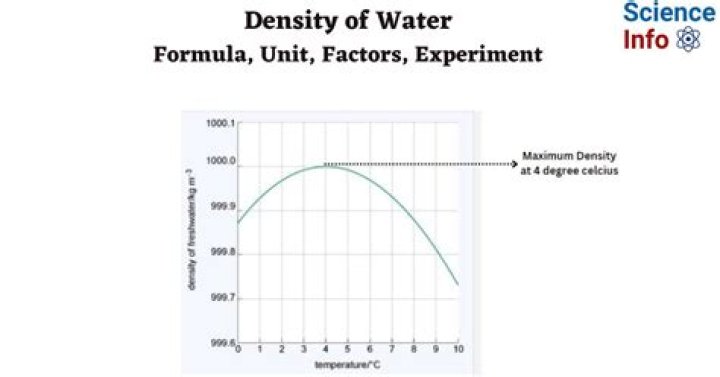
As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart. … Thus, the density of water is a maximum at 4 °C.
Why is water more dense at 4 degrees Celsius liquid than it is as ice solid )?
Density increase as the temperature decreases. Below 4 deg C, however, the density decreases again. … This is the reason why liquid water is more dense than solid water. The bonds in water break more slowly as temperature decreases and the structure tend to trap fewer extra water molecules.
What is the maximum density of water at 4 degrees Celsius?
Detailed Solution. The maximum density of water is 103 kg m-3 at 4oC temperature.
What is the density of water at 4 degrees?
Actually, the exact density of water is not really 1 g/ml, but rather a bit less (very, very little less), at 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit).At which temperature does water have its greatest density?
In almost all substances the atoms and molecules move closer together as they get colder. They then solidify. Water, however, attains its greatest density at four degrees Celsius because the water molecules are packed closest together at this temperature.
What happens to water when its temperature reaches 4 degrees C?
Water is one of the few exceptions to this behavior. When liquid water is cooled, it contracts like one would expect until a temperature of approximately 4 degrees Celsius is reached. After that, it expands slightly until it reaches the freezing point, and then when it freezes it expands by approximately 9%.
What happens to water at 4 degrees Celsius?
4 degrees C turns out to be the temperature at which liquid water has the highest density. If you heat it or cool it, it will expand. The expansion of water when you cool it to lower temperatures is unusual, since most liquids contract when they’re cooled.
What is the density of 4?
DensityName#1.848 g/ccBeryllium41.873 g/ccCesium552.07 g/ccSulfur162.26 g/ccCarbon6What is the volume of water at 4 degree Celsius?
TemperatureSpecific volume (0-100°C at 1 atm, >100 °C at saturation pressure)0.11.000150.1198111.000100.1198141.000030.11982101.000300.11979
Why does water have high density?It’s All About Hydrogen Bonds When water freezes into ice, it crystallizes into a rigid lattice that increases the space between molecules, with each molecule hydrogen bonded to 4 other molecules. … “Why Is Water More Dense Than Ice?” ThoughtCo, Aug. 27, 2020,
Article first time published onAt what temperature the density of water is maximum and why?
An especially notable irregular maximum density is that of water, which reaches a density peak at 4 °C (39 °F). This has important ramifications in Earth’s ecosystem.
At what temperature is the water at its least dense?
Water is densest at 3.98°C and is least dense at 0°C (freezing point). Water density changes with temperature and salinity.
How does water temperature affect density?
When the water is heated, it expands, increasing in volume. … The warmer the water, the more space it takes up, and the lower its density. When comparing two samples of water with the same salinity, or mass, the water sample with the higher temperature will have a greater volume, and it will therefore be less dense.
Why is the critical temperature of water greater than that of most other substances?
Since water is governed by stronger forces of attraction (H-bond) than H2S (dipole-dipole), more energy is needed to break apart water molecules into irreversible gaseous state beyond critical temperature and pressure.
Why does water freeze at 4 degrees?
Below 4° Celsius, water becomes less dense as it gets colder, causing water about to freeze to float to the top. … And because the same mass of molecules takes up more space when frozen, ice is less dense than liquid water. For this same reason, water below 4° Celsius becomes increasingly less dense as it gets colder.
At what temperature does water expand?
Between 32 and 40 degrees Fahrenheit (0 and 4 degrees Celsius), the melted water actually contracts as the temperature rises. Beyond 40 F (4 C), it starts to expand again.
Why is 4c the critical temperature?
Why is 4°C the critical temperature? As a result of hydrogen bonding, water expands as it solidifies. At 4°C water acts like other liquids, expanding as it warms and contracting as it cools. However, below 4°C water begins to freeze as more and more hydrogen molecules are moving to slowly to break the hydrogen bonds.
Which expands the greatest when heated through the same temperature?
Question 27 Q28) State which expands more, when heated to the same temperature: solid, liquid or gas? Solution: Gases expand much more than the liquid and the solids. Like liquids, the gases do not have a definite shape, so they also have only the cubical expansion.
Why does water expand when heated?
2.1 A was heated. An increase in temperature caused the water molecules to gain energy and move more rapidly, which resulted in water molecules that are farther apart and an increase in water volume. … When water is heated, it expands, or increases in volume.
How do you find the density of water at 4 degrees Celsius?
1Kg/m3.
Why is the volume of water minimum at 4 C?
When water is cooled, the density of water gradually increases till 4o C. At 4oC, water exhibit maximum density and minimum volume. On further cooling down, water expands and the density of water decreases from 4oC to 0oC.
Is more density heavier?
If a substance has a higher density, it is heavier. Likewise a lighter density means it is much lighter. For example, air has a density of approximately 1.225kgm3 .
Which element has highest density?
As it turns out, either of two elements can be considered the element with the highest density: osmium or iridium. Both osmium and iridium are very dense metals, each weighing approximately twice as much as lead.
At what temperature is water most dense quizlet?
Water is the most dense at 4 degrees celsius, just prior to freezing. (Cold water in a pond will stay at the bottom.)
When the temperature of water decreases from 4 degree Celsius to 2 degree Celsius will the level of water rise or fall in the cylinder?
Water is at its maximum density at 4.0°C . This means that if you increase the temperature – OR decrease the temperature of the water – the density will drop. Because the mass remains constant , the volume of the water must increase if the density reduces.
Why water has more density than ice?
The “stuff” (molecules) in water is more tightly packed than in ice, so water has greater density than ice. … As water freezes it expands. So, ice has more volume (it takes up more space, but has less density) than water.
Why is cold water more dense than warm water?
Cooling a substance causes molecules to slow down and get slightly closer together, occupying a smaller volume that results in an increase in density. Hot water is less dense and will float on room-temperature water. Cold water is more dense and will sink in room-temperature water.
Which has a greater effect on density of water temperature or pressure?
The density of water increases as the salinity increases. The density of seawater (salinity greater than 24.7) increases as temperature decreases at all temperatures above the freezing point. The density of seawater is increased by increasing pressure.
Why does a larger volume of water take longer to cool?
Hypothesis: If the volume of the water is increased, then the rate of cooling will be slower because there are more molecules in greater volume than less volume. Thus, it will take more time to lose the heat energy from all the molecules.
What happens above the critical temperature?
Above the critical temperature, the molecules have too much kinetic energy for the intermolecular attractive forces to hold them together in a separate liquid phase. Instead, the substance forms a single phase that completely occupies the volume of the container.
Which of the following gas has highest critical temperature?
Water vapours i.e., H2O(g) molecules can be liquefied most easily due to presence of intermolecular hydrogen bonding. Therefore, they have maximum critical temperature .