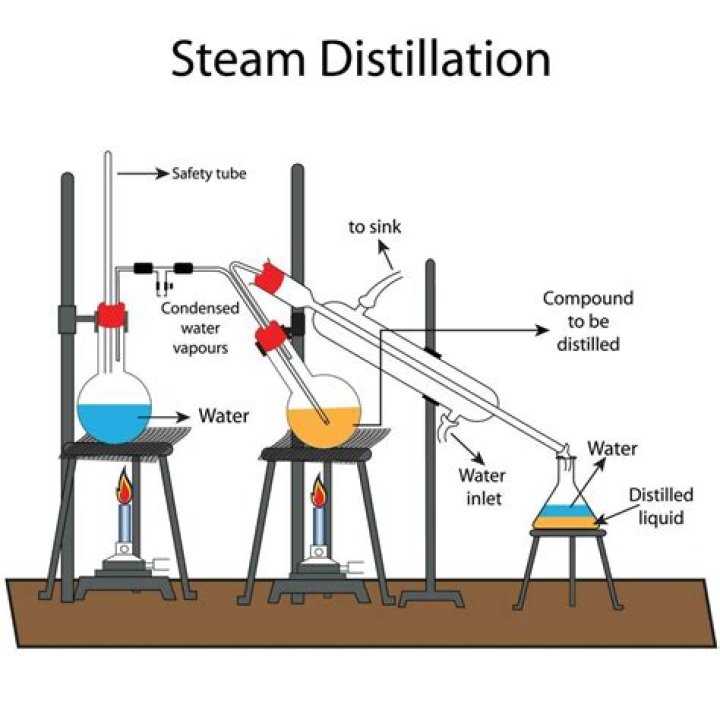
Steam or water is added to the distillation apparatus, lowering the boiling points of the compounds. The goal is to heat and separate the components at temperatures below their decomposition point.
Why is steam distillation used for the isolation of essential oils?
Steam distillation, the method used in this lab for essential oil extraction, takes advantage of the volatility of a compound to evaporate when heated with steam and the hydrophobicity of the compound to separate into an oil phase during condensation.
What can be steam distilled?
Steam distillation is used to separate temperature-sensitive organic compounds such as aromatic substances. It can be used to extract oils from natural products, such as eucalyptus oil, citrus oils, or other natural substances derived from organic matter.
Why steam distillation is more suitable for the compounds which have high boiling point?
Steam Distillation The goal is to heat and separate the components at temperatures below their decomposition point. The advantage of steam distillation over simple distillation is that the lower boiling point reduces decomposition of temperature-sensitive compounds.Why is steam distillation used to isolate limonene?
Limonene is a high-boiling liquid (bp763 = 175.5-176 °C) which decomposes under the high heat needed to bring it to a boil. Steam distillation is a much gentler way to obtain essential oils, as it allows the distillation of co-mixture of oil and water at a boiling point less than 100 °C.
Why is boiling point lower in steam distillation?
This technique is used for separating substances which are immiscible with water, volatile in steam & having high vapour pressure at the boiling temperature of water. Hence the mixture of two immiscible liquid will boil at a lower temperature than the normal boiling point. …
What is the advantage of steam distillation Mcq?
Explanation: Steam distillation has very long extraction times, requires high energy consumption and there is no need for subsequent separation steps. It generates organic solvent-free products.
How is steam distillation performed?
Steam distillation is carried out by passing dry steam through the plant material whereby the steam volatile compounds are volatilized, condensed and collected in receivers. … Distillation is carried out with low pressure steam which replaces the volatile compounds from the intact plant material.How is steam distillation different from simple distillation?
Different. Simple distillation is powered by a direct heat source usually of over 100°C. In steam distillation the heat source is steam passing through the medium being distilled. It is therefore usually performed at much lower temperature except where the steam is under pressure.
Why is the distillate cloudy in steam distillation?The newly condensed steam distillate appears cloudy because it is a mixture (really an emulsion) of water and eugenol, which is not water-soluble.
Article first time published onWhat is the name of the extraction method used to extract lemongrass and spices?
It is also used to produce perfumes, avours, detergents, and pharmaceuticals. The method found to be the most suitable for the extraction of lemongrass essential oil is steam distillation, since it allows obtaining the oil without altering product quality. The chemical composition of the essential oil of C.
What is R limonene?
Limonene is a colorless liquid aliphatic hydrocarbon classified as a cyclic monoterpene, and is the major component in the oil of citrus fruit peels. The D-isomer, occurring more commonly in nature as the fragrance of oranges, is a flavoring agent in food manufacturing. … Racemic limonene is known as dipentene.
What distillation is used for?
Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or in the separation of two or more liquids having different boiling points, as in the separation of gasoline, kerosene, and lubricating oil from crude oil.
Why distillation is applied to some mixtures?
Distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. To separate a mixture of liquids, the liquid can be heated to force components, which have different boiling points, into the gas phase.
What substances Cannot be separated by steam distillation?
a,b and c are steam volatile, therefore, can be separated by steam distillation and they are insoluble in water. Glycerol decomposes at its boiling point as well as it is soluble in water. It cannot be separated by steam distillation, rather it is purified by distillation under reduced pressure.
Why is steam distillation used for eugenol?
Eugenol has a high boiling point (254 oC), and many organic compounds decompose at such high temperatures. Steam distillation allows eugenol to be distilled at a much lower boiling point (< 100 oC), thus minimizing the potential for decomposition.
Which is purified by steam distillation?
Aniline is purified using the process of steam distillation because aniline is steam volatile and it is insoluble in water.
Why is the boiling point of steam distillation always less than 100?
Steam distillation is also used for high boiling point substances that decompose before the boiling point is reached. … Because the two are immiscible, they will steam distill at a temperature around 90°C, less than 100°C (the boiling point of water) and well below 218 °C.
How do you separate eugenol and eugenol acetate?
D Extract organic layer 3x with aq. NaOH (1.5 M) to separate eugenol from eugenol acetate. Eugenol (a phenol) gets deprotonated by NaOH and becomes a salt which moves to aq. layer.
What is the purpose of the boiling chips in distillation?
Boiling chips are frequently employed in distillation and heating. When a liquid becomes superheated, a speck of dust or a stirring rod can cause violent flash boiling. Boiling chips provide nucleation sites so the liquid boils smoothly without becoming superheated or bumping.
What is eugenol classified?
Eugenol is a phenylpropanoid formally derived from guaiacol with an allyl chain substituted para to the hydroxy group. It is a major component of clove essential oil, and exhibits antibacterial, analgesic and antioxidant properties. … It is a phenylpropanoid, a monomethoxybenzene and a member of phenols.
Where is oleoresin used?
Use. Most oleoresins are used as flavors and perfumes, some are used medicinally (e. g., oleoresin of dry Cannabis infructescence). Oleoresin capsicum is commonly used as a basis for tear gases. There are also uses known in the manufacture of soaps of cosmetics, as well as coloring agents for foods.
What is the principle behind hydro distillation?
In this case, the raw materials are immersed in boiling water. The volatile compounds are carried away with the water vapor and collected after condensation and decantation, as in steam distillation. Hydrodistillation is generally carried out at atmospheric pressure.
What is oleoresin of ginger?
Ginger Oleoresin is a prepared oleoresin. It is produced by extraction of the dried and unpeeled rhizome of Zingiber Officinale, ground to a moderately coarse powder. The removal of the last few percents of solvent is a problem which has yet to be solved satisfactorily.
What is D-limonene used for?
Being a solvent of cholesterol, d-limonene has been used clinically to dissolve cholesterol-containing gallstones. Because of its gastric acid neutralizing effect and its support of normal peristalsis, it has also been used for relief of heartburn and gastroesophageal reflux (GERD).
Where is citral from?
Citral is a main component of citrus fruit’s peel oil. It is especially found in orange peel. Citral is a mixture of neral and geranial which are monoterpene aldehydes (Maarse, 1991). Citral has been applied to food, cosmetics, and beverages as a natural ingredient for its passionate lemon aroma and flavor.
What is D-limonene made of?
D-limonene is a compound sourced from the peel of citrus fruits, including include oranges, mandarins, limes, and grapefruit.
Why does distillation produce pure water?
This method works because water has a much lower boiling point than salt. When the solution is heated, the water evaporates. It is then cooled and condensed into a separate container. The salt does not evaporate and so it stays behind.
Where is simple distillation used?
Uses. Simple distillation is the primary method used for purifying drinking water on both a large and small scale. There are many models available for purifying water on a smaller scale. Simple distillation is also a primary method for the purification of ethanol into fuel grade alcohol.
Why is an ice bath used in distillation?
Since the vapor pressure of water at 20 °C is roughly 2.3 kPa, then 2.3/101.325 or 0.023 would be the fraction of water that would not condense and would pass out of the condenser. This is why the distillate is frequently chilled in an ice bath during the distillation.
What is example of steam distillation?
Steam distillation is often employed in the isolation of essential oils, for use in perfumes, for example. In this method, steam is passed through the plant material containing the desired oils. Eucalyptus oil, camphor oil and orange oil are obtained by this method on an industrial scale.